Paper 10
Quantification of messenger RNA and Analysis of mRNA-
Translated Protein Products Induced by IBA During
Different Stages of Root Formation.
G. R. KANTHARAJ* and G. PADMANABHAN**
* Dept. of Botany, The National Collage, Bangalore – 560 004, India. (Correspondence address)
Dept. of Biochemistry, Indian Institute of Science, Bangalore - 560 012,
India
INTRODUCTION
The results presented in Chapter I indicate that some of the early proteins synthesized in response to IBA treatment play a vital role in adventitious root initiation. The results have indicated that inhibition of the very early protein synthesis (30 min) results in an inhibition of RNA synthesis and thus of the early proteins (2-12 hrs) coded for by the early mRNAs synthesized in response to IBA. The enhanced radio-labeling of total RNA and poly (A)-containing RNA, in response to IBA are taken as a measuring of the effect of the hormone to the rate of RNA synthesis. The increased messenger RNA content can also be quantified in terms of the translatable messenger activity of the RNA isolated from IBA-treated hypocotyls in a cell-free system.
Hence, in the present study, total and poly(A)-containing RNA have been isolated from hypocotyls with and without hormone treatment and these RNA fractions have been translated in a wheat germ cell-free system. In addition to quantifying the messenger RNA content, the labeled proteins synthesized in the cell-free system have been analyzed using SDS-gel electrophoresis.
MATERIALS AND METHODS
Materials:
[3H] – Leucine (105 Ci / mmole) was purchased from Radiochemical Centre, Amersham, bucks, England. Oligo-dT-cellulose was purchased from Collaborative Research Inc., Waltham, Massachusetts, U.S.A. All other chemicals used were of analytical grade.
Methods:
Treatment of the hypocotyls:
As described in Chapter I, hypocotyls segments were placed in Hoagland’s medium containing IBA (30 mg/ml). After 30 min exposure, the hypocotyls were thoroughly washed with water and transferred to flasks containing fresh hormone-free medium. The hypocotyls were removed at different intervals of time and used for RNA isolation.
Isolation of total and poly (A) – containing RNA:
The methods for isolation of total RNA as well as poly (A)-RNA have already been described in Chapter I. Total RNA isolated from 50-100 fm of hypocotyls tissue was washed with 2 volumes of 70% ethanol containing 2M Potassium acetate and then thrice with 3M sodium acetate (pH 6.8). The RNA was reprecipitated with ethanol and the pellet was used for translation. Poly (A)-containing RNA was isolated using two cycles of oligo-dT-cellulose chromatography.
Translation of RNA in the wheat germ cell-free system:
The procedure of Roberts and Patterson [234] was used with some modifications. In these experiments glassware was washed in acid and baked overnight at 200oC and all aqueous solutions were autoclaved in order to eliminate inadvertent RNase contamination.
Preparation of S-30 extract:
Wheat germ (1 gm) was ground with 4 ml of buffer A (containing 200 mM HEPES, pH 7.5, 1 mM magnesium acetate, 100mM KC1, 2mM CaCl2 and 6 mM b-mercaptoethanol) and 1 gm sand in a pre-cooled mortar and pestle. The homogenate was centrifuged at 15000xg for 10 minutes. The supernatant (1.5 ml) was fed onto a Sephadex G-25 column equilibrated with buffer B (containing 20 mM HEPES, pH 7.5, 5 mM magnesium acetate, 120 mM KCl and 6 mM b-mercaptoethanol). The void volume was collected and peak fractions (A260) were pooled and centrifuged at 15000xg for 10 minutes. The supernatant was used as the S-30 source.
Assay procedure:
The assay mixture in a total volume of 0.1 ml contained : HEPES, 20 mM; ATP, 1 mM; GTP, 20 mM; phosphocreatine, 8 mM; magnesium acetate, 3 mM; potassium chloride, 100 mM; amino acid mixture without leucine, 25mM each; DTT, 2 mM; [3H] – leucine 105 Ci / mmole), 5 mCi; S-30 fraction, about one A260 unit and RNA (optimum concentration). The mixture was incubated at 25oC for 1 hr and 5 ml aliquots were transferred to filter paper discs to be processed for measurement of radioactivity in the TCA precipitable proteins as described in experimental methods of Chapter I
SDS-Gel analysis of labeled proteins synthesized in the cell-free system derived from the wheat – germ:
The labeled protein products synthesized in the wheat germ cell-free system in response to RNA isolated from hypocotyls with and without hormone treatment, were precipitated with cold trichloroacetic acid TCA (10% in final concentration) and then washed successively with cold TCA (5%), ethanol-ether (3:1), and finally with ether. The pellet was dissociated in 2% SDS containing 5% b-mercaptoethanol, 10mM Tris – HCL (pH 6.8) and 10% glycerol and heated in a boiling water bath (100oC) for 2 min and analyzed by SDS-gel (8%) – electrophoresis as described in Chapter I.
The gels were stained with Coomassie blue-R and cut into 1 mm slices. The slices were digested with 0.5 ml of 30% hydrogen peroxide at 60oC and radioactivity was measured using 0.5% PPO in Triton-toluene (1:2, v/v) mixture.
RESULTS
Isolation of poly (A)-RNA:
The poly (A)-containing RNA elution profile from oligo-dT-cellulose column is given in Figure 7. Poly (A)-containing RNA was obtained in yields of O.4-O.6% of total RNA.
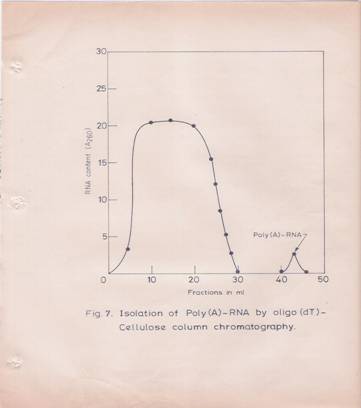
Fig7: Isolation of Poly (A)-containing RNA by Oligo dT-cellulose Column Chromatography,
Total RNA was dissolved in O.O1 M Tris-HCL (pH 7.5) buffer containing O.5M NaCl and 0.5% SDS (High-Salt Buffer). About 250 A260 units of total RNA from the hypocotyls tissue was loaded on to a 2.5 ml oligo dT-column (O.5 gm of oligo dT-cellulose), which was equilibrated with the above high-salt buffer. The column was washed first with the equilibrating buffer followed by high salt buffer containing 0.3M NaCl without SDS. Poly (A)-containing RAN was eluted with O.O1M Tris-HCL (pH 7.5) buffer.
Determination of various factors for optimal translation of RNA in vitro in the wheat germ system:
Various parameters for the optimal translation of total and poly (A)-containing RNA in the wheat germ system were established. The translational response to exogenous RNA was optimal at 3.25 mM Mg24 and 100 mM K4 concentrations (figures 8 and 9).
A linear response with increasing RNA concentration (50 mg/ml) for poly (A) – RNA and 400 mg/ml for total RNA) in terms of protein synthesis was observed (Figures 10 and 11). The reaction rate was linear up to 60 min (Figure 12). Under these conditions, a stimulation of 15-20 folds over the endogenous protein synthesis was observed.
Quantification of messenger RNA content:
In the wheat germ cell-free system, quantification of translatable activity of the RNA is possible, since the amount of protein synthesized is proportional to the amount of RNA added up to a certain concentration of the latter. Therefore, an estimate of the messenger content was attempted by comparing the translation efficiency of total RNA preparations from control and IBA-treated hypocotyls. The results (Table 10, Figure 13) indicate that at the same amount of total RNA (200 mg.ml), the preparations from IBA-treated hypocotyls, elicit a higher amount of protein synthesis as expressed in terms of [3H]-leucine incorporated into proteins (cpm/A260 units per hr).

Fig. 8: Effect of Mg2+ concentration at 100 mM K+ on translation of hypocotyls RNA in the wheat germ cell-free system.
The in vitro translational mixture in a final volume of 100 ml contained (in final concentration) 100 mM KCL, 300 mM spermidine, 4 mg of poly (A) – RNA and other components. The reaction was carried out as described in the experimental part. Increasing concentration of magnesium was added to the reaction mixture and incubated for 60 minutes. The incorporation of [3 H] – leucine into TCA-precipitable protein was determined. The values given are the mean of two replicates.

Fig. 9: Effect of potassium concentration at 3.25 mM Mg2+ on the translation of hypocotyls RNA in the wheat germ cell-free system.
The experimental details are as given in Fig. 8. The Mg2+ concentration used was 3.25 mM. The values given are the mean of two replicates.

Fig. 10: Effect of poly (A)-RNA concentration on protein synthesis in vitro in the wheat germ cell-free system.
The reaction mixture in a total volume of 100ml contained 3.25 mM of Mg++ acetate, 100 mM KCL, 300 mM spermidine and other components, and translation was carried out as described in experimental methods. The effect of increasing concentrations of poly (A)-RNA was tested. The values given are the mean of two replicates.

Fig. 11: Effect of total RNA concentration on protein synthesis in vitro in the wheat germ cell-free system.
The experimental details are as given in Fig.10 except that the total RNA was used in the place of poly (A)-RNA. The values given are the mean of two replicates.

Fig. 12: Time course of protein synthesis in the wheat germ cell-free xylem in the presence of added poly (A)-containing RNA.
The incubation mixture (100 ml) containing 4 mg of poly (A)-RNA was incubated at 250C for different time intervals. The incorporation of [3H]–leucine into TCA precipitable protein was determined. The values given are the mean of two replicates.
Table 10: Effect of IBA-treatment on translatable RNA activity in the hypocotyls segment of Phaseolus vulgaris .L.
_______________________________________________________
RNA sources [3H]-Leucine incorporated into TCA perceptible
Protein (cpm/A260)
___________________________________________
Experiment 1 Experiment 2
_______________________________________________________
Control 0 hr 45500 54300
IBA ½ hr 69520 77500
IBA 6 hr 77700 90500
IBA 12 hr 112000 120800
IBA 24 hr 136100 142000
IBA 48 hr 165300 176000
_______________________________________________________
Total RNA was isolated from the hypocotyls tissue at various time intervals of IBA treatment. Equal amount of total RNA was translated in cell-free system derived from wheat germ. [3H]-Leucine incorporation into TCA precipitable protein was determined. The results are expressed in terms of leucine incorporated into TCA precipitable protein (cpm/A260 of RNA translated). Each value is the mean of two replicates. Results of two independent experiments have been presented.

Fig 13: IBA induced translatable mRNA-content at various stages of adventitious root initiation.
Total RNA was isolated at various stages of adventitious root initiation. Equal amounts RNA were translated in vitro system and the [3H]-leucine incorporated into TCA precipitable protein was plotted against time at which the RNA preparation was obtained. Each value is the mean of two replicates.
Terms of [3H]-leucine incorporated into the protein (cpm/A260) unit/hr). Total RNA preparations isolated from the hypocotyls tissue after 24 and 48 hr of IBA treatment, stimulate protein synthesis by 3 and 4-fold more than that of the corresponding preparations from untreated tissue.
EFFICIENCY OF TRANSLATION
An intriguing result is obtained when the translation efficiency of the different poly (A)-containing RNA preparations have been compared. The results (Table 11) indicate that poly (A)-containing RNA concentration of 50 mg/ml. However this difference is nearly constant for all the preparations with the poly (A)-containing RNA from IBA treated tissue (regardless of the duration of treatment) being 1.5 times more active than that of control.
ANALYSIS OF TRANSLATED PRODUCTS IN VITRO:
Finally, the labeled translation products were analyzed by SDS-gel electrophoresis and the radioactive profiles of the proteins are given in Figures 14 and 15. The total and poly (A)-containing RNA preparations isolated from hypocotyls after 6 and 12 hrs of IBA treatment were used in these studies. The profiles of the labeled proteins synthesized in response to the added RNA from control and IBA treated.
Table 11: Effect of IBA-treatment on the translatable activity of poly (A)-containing RNA.
________________________________________________________________
RNA sources [3H]-Leucine incorporated into TCA precipitable
Protein (cpm/A260)
___________________________________________
Experiment 1 Experiment 2 Experimet3
________________________________________________________________
Control 0 hr 47200 39400 46000
IBA ½ hr 62000 61500 60500
IBA 6 hr 64800 64300 63500
IBA 12 hr - 70000 68000
Poly (A)-RNA was isolated from the hypocotyls at various stages of development after IBA treatment and 5 mg of RNA from each sample was translated in cell free system derived from wheat germ as described in the experimental part. The results are expressed in terms of [3H]-leucine incorporated into TCA perceptible (cpm/s mg of poly (A)-RNA translated). Each value is the mean of two replicates and the results obtained in three independent experiments are given.

Fig.14: Radioactivity profile of poly (A)-RNA translated products in the cell-free system derived from wheat germ.
Poly (A)-RNA isolated at different stages after IBA treatment was translated and the labeled products were analyzed by SDS-gel electrophoresis. The profiles represent the labeled protein products derived from poly (A)-RNA isolated at (A) 0 hr; (B) 6 hr; (C) 12 hr, after IBA treatment.

Fig. 15: Radioactivity profile of total RNA translated products in the cell-free system derived from wheat germ.
The details are as described in fig. 14 except that total RNA was used in the translation. The profiles represent the labeled protein products derived from total RNA isolated at (A) 0 hr; (B) 6hr and (C) 12 hr after IBA treatment.

Fig. 16: SDS-slab gel electrophoresis of in vitro translated products of RNA obtained from hypocotyls.
The translated protein products were electrophoresed on slab gels. The gels were stained with Coomassie blue.
Lane 1 – Standard proteins.
Lane 3, 4, 5 and 6 – represent preparations after translation of RNA isolated at 0, ½, 6 and 12 hr after IBA treatment.
Lane 2 – represents the endogenous control.
Tissues show certain significant differences. The conspicuous changes brought about by IBA treatment appear to be the synthesis of messenger RNAs for certain proteins in the molecular weight range of 80,000 to 1, 75,000 daltons and certain low molecular weight proteins, as well as an enhanced labeling of protein of molecular weight around 55,000 daltons. The enhanced labeling of the 55,000 molecular weight protein is more conspicuous in the pattern obtained with poly (A)-containing RNA.
The Coomassie blue-stained SDS-gel photograph of the products of in vitro translation is given in the fig. 16. It may be pointed out that proteins stained would be essentially the wheat germ proteins. Since, an enhanced labeling of proteins in the low molecular weight region was also seeing in the case of translated products of RNA from IBA-treated hypocotyls, the possibility of in vitro protein degradation contributing artifact to this situation was considered. It was felt that if such degradation was taking place, it might as well be reflected in the degradation of wheat germ proteins. The staining patterns in figure 16 are identical in all the slots (with or without added RNA)
DISCUSSION
The increase in messenger RNA content during hormone induced differentiation process has been well documented for example, GA3 induces messenger RNA synthesis
For a-amylase in barley aleurone cells [90-92,231,232]. An increase in messenger RNA synthesis at the early stages of hormone treatment (3-12 hr) in pea epicotyl and soybean hypocotyls is reported, although the nature of the protein products is not clear [72,151,196] except for cellulase [79].
In the present study, two types of IBA-mediated effect on translatable messenger RNA are noticed. After 6 hours of IBA treatment, translatable messenger RNA activity of total RNA increases by nearly 3 fold. The increase is between 3 and 4-fold after 24 and 48 hours of IBA treatment respectively. Thus it can be concluded that the hormone increases the messenger RNA content in hypocotyls segments.
The finding that poly(A)-containing RNA from IBA-treated hypocotyls is 1.5 fold more active than that from the control tissue, indicates that hormone has an effect on the quality of the messenger fraction. The reason from this effect of IBA is not apparent. However, it could be due to the effect of hormone treatment on the secondary structure of RNA, ribonuclease activity, etc., A similar finding is reported in the case of messenger RNA fractions isolated from thioacetalamide treated rat livers [233].
The IBA-induced increase in the translatable activity of poly (A)-containing RNA remains constant for the RNA preparations isolated at different interval of times. The translatable activity of total RNA obtained from 48 hr treatment, however, increases by 4 fold; thus, the hormone brings about an increase in the quantity as well as quality of the messenger RNAs. An exact quantification of messenger sequences would need C-DNA hybridization studies with the RNA population. The higher translatable activity of poly (A)-containing RNA from IBA- treated hypocotyls may also , in part, explain the effect of the hormone in eliciting enhanced protein synthesis as early as 30 minutes (chapter I) without the mediation of enhanced RNA synthesis.
The analysis of protein products coded for by the RNA isolated from IBA – treated hypocotyls, in the wheat germ cell free system, reveals that the effects are quantitative as well as qualitative. In particular, there is an enhanced synthesis of proteins in the molecular weight ranges of 80,000-1, 75,000 55,000 and 12,000-5,000. The enhanced labeling of the low molecular weight proteins, however, does not appear to be due to artifactual degradation. The early proteins synthesized perhaps play a vital role in adventitious root initiation and further studies to characterize the 55,000 molecular weight protein are reported in the following chapters.
SUMMARY
1. Various parameters for the optimal translation of total and poly (A)-containing RNA in cell-free protein synthesizing system from wheat germ have been established.
2. The messenger RNA content increases by 2-4 fold over a period of 6 to 48 hours after IBA treatment.
3. Poly (A)-containing RNA preparations from IBA treated hypocotyls uniformly exhibit 1.5 fold higher translational efficiency than the RNA from control tissue irrespective of the duration of IBA treatment.
4. Total and poly(A)-RNA from IBA treated hypocotyls on translation manifest a higher rate of synthesis of high molecular weight proteins (80,000 to 1,75,000 daltons); a protein of 55,000 daltons and low molecular weight proteins (12,000 – 5,000 daltons).
CHAPTER III
Identification and Characterization of Indole Butyric Acid-Induced Membrane Protein of Molecular Weight 55,000 Daltons
INTRODUCTION
The role of microtubules and microfilaments in cellular metabolism and organogenesis in plants is very well documented [9, 235]. Rizogenesis in developing Fucus zygotes can be inhibited by the application of colchicine and cytochalasin B. In the petioles of French beans, colchicine is found to inhibit root formation [236]. The above observations suggest that microtubules and microfilaments have a definite role in organogenesis. Tubulin and actin are important protein constituents of the cytoskeletal structures of a variety of cells [240]. Recent studies also implicate tubulin as a component of microtubular network in morphogenesis and differentiation [237 –239, 245].
Tubulin exists in the form of heterodimers consisting of a and b subunits of mol, wt, 110,000 [16]. The detection of significant labeling of the 55,000 mol, wt. Protein in the translation products of RNA isolated from IBA-treated hypocotyls indicated the possibility that the hormone could be inducing the synthesis of tubulin and its messenger RNA, which may play a vital role in the process of adventitious root initiation. In the following investigations efforts are made to examine the protein profiles of the IBA treated hypocotyls tissue.
MATERIALS AND METHODS
Materials:
Phosphocellulose was purchased from Sigma Chemicals Company, U.S.A. All other chemicals used were of analytical grade.
Methods:
Preparation of soluble and membrane proteins from hypocotyl tissue:
IBA treated and control hypocotyls segments were used for the isolation of soluble and membrane fractions. The tissue (30-40 Gms) was homogenized in). O.O2M sodium phosphate buffer (pH 7.0) containing O.25M sucrose and 10 mM PMSF in a precooled all glass-mortar and pestle. The homogenate was filtered through two layers of cheese cloth and the filtrate was centrifuged at 10,000 rpm for 10 min. The post-mitochondrial supernatant was again centrifuged at 42,000 rpm for 90 min. The supernatant (soluble fraction) was carefully removed. The membrane pellet was once rinsed with the homogenizing buffer and suspended in O.5 ml of the same.
Effect of cytochalasin B and colchicines on root initiation:
Hypocotyl segments (4-6 cm length) were treated with IBA (30 mG/ML) FOR 30 MIN. Then the segments were washed and placed in fresh nutrient medium. At different time intervals cytochalasin B (10 mg/ml) or colchicines (2.5 mg/ml) was added. After incubating for 6 hr, segments were removed and washed repeatedly in water and placed in fresh Hoagland’s medium. At the end of 72 hr, the roots formed were counted.
Isolation of tubulin from rat brain:
The isolation of tubulin from rat brain was carried out according to the methods of Weingarten et al with certain modifications. Adult rat brains were homogenized in 2 volume of PMS buffer (pH 6.8) (O.1M sodium pyrophosphate, O.O1M magnesium chloride and O.1M sucrose), and the homogenate was centrifuged at 10,000 rpm for 10 min. The supernatant was then spun at 42,000 rpm for 60 min in a preparative ultracentrifuge. The post-microsomal supernatant was carefully removed, mixed with an equal volume of Phosphocellulose (5 gm) equilibrated with PMS buffer (pH 6.8) and stirred slowly for 60 minutes. Then the mixture was centrifuged at 15,000 rpm for 15 min. The supernatant was removed and the Phosphocellulose pellet was washed successively with PMS buffer (pH 6.8) containing O.15M sodium chloride three times. The Phosphocellulose pellet was again washed three times with PMS buffer (pH 6.8) containing O.2M sodium chloride. Tubulin bound to Phosphocellulose was eluted with PMS buffer (pH 6.8) containing O.3-O.4M sodium chloride, by repeated centrifugation procedure. The eluate was dialyzed overnight against O.2M sucrose solution. The dialyzed tubulin was concentrated with sucrose and stored at –20oC.
Isolation of tubulin from the hypocotyls segments of Phaseolus Vulgaris:
Plant tubulin was extracted by the same procedure as described for rat brain except that the PMS buffer used for homogenization and washing contained in addition 1 Mm DTT and 10mM phenylmethylsulphonyl fluoride. The post-mitochondrial supernatant was solubilized with the addition of deoxycholate to a final concentration of 1% and used for the isolation of tubulin. The phosphor-cellulose used for adsorbing tubulin was also equilibrated with PMS buffer (pH 6.8) containing O.15M NaCl and 1% deoxycholate. After repeated washing (three times each) in PMS buffer containing O.15M NaCl and O.2M NaCl, tubulin was eluted with PMS buffer containing O.3M-O, 4M NaCl. The eluate was dialyzed overnight against O.2M sucrose, and then the tubulin was concentrated with sucrose and stored at –20oC.
Characterization of tubulin antibody:
Preparation of antibody to the rat brain tubulin:
Male albino rabbits (New Zealand strain) weighing about 2 Kg were injected with 2 mg of purified rat brain tubulin in Freund’s complete adjuvant. A booster dose was given after 21 days of the first injection. After seven days of the booster does, rabbits were bled from the marginal veins of the ear. The serum was separated from the agglutinated blood by centrifugation at 10,000 rpm for 10 min and sodium azide (O.O2%) was added to the serum and stored at –20oC.
Preparation of g - globulin:
The g - globulin fraction was prepared from the antiserum according to the methods of Campbell el al. (241). The antiserum was diluted with an equal volume of 0.2M sodium phosphate buffer (pH 7.5). Solid ammonium sulfate was added to 40% saturation and kept at –4oC for 1 hr. Then, it was centrifuged at 10,000 rpm for 10 min. The precipitate was dissolved in 0.2M sodium phosphate buffer (pH 7.5) and dialyzed against the same buffer and passed through DEAE-cellulose column which was pre-equilibrated with the above buffer. The g - globulin fraction, that came out in the column volume was collected and reprecipitated by the addition of solid ammonium sulfate to 40% saturation. The precipitate was collected by centrifugation and dissolved in 0.02M Tris-HCl (pH 7.4) containing 0.15M sodium chloride and dialyzed against the same buffer. After adding sodium azide to a final concentration of 0.02%, the g - globulin fraction was stored in small aliquots at –20oC.
Ouchterlony double diffusion analysis:
Ouchterlony plates were prepared according to Thomas et al. [242]. Agarose (0.9%) was dissolved in 1.0M glycine buffer (pH 7.4) containing 0.08M sodium chloride, 0.15M sodium azide; heated to boiling and pored into Petri dishes (9 cm in diameter) to 3 mm thickness. Circular wells of required dimensions were made. The antibody preparation was put into the central well and the antigen in the peripheral, wells. The plates were left at room temperature for 1-2 hr and incubated at –4o C for 48 hrs. After the precipitin lines had developed, the plates were washed repeatedly with 0.05M potassium phosphate buffer (pH 8.0) containing 0.3M sodium chloride and photographed.
Quantitative immunoprecipitation:
Membrane fraction from the hypocotyls segment was isolated as described earlier. The membrane fraction was solubilized in 0.02M Tris-HCl (pH 7.4) buffer containing 0.15M NaCl, 10 mM PMSF and 1% deoxycholate. Increasing concentrations of membrane proteins were dispensed into immunoprecipitation tubes and purified antibody (550 mg protein) rose against rat brain tubulin, was added to each tube. The final volume was made up to 0.5 ml with 0.02M Tris-HCl (pH 7.4) buffer. Then the tubes were incubated at 27o C for one hour and at 4oC overnight. The immunoprecipitates were collected by centrifugation at 2500 rpm for 15 min. The immunoprecipitates were washed with 0.5 ml of 0.02M Tris-HCl (pH 7.4) buffer containing 0.15M NaCl. Separate blanks for membrane proteins and antibodies were also processed. The final immunoprecipitates were dissolved in 0.2 ml of 0.01N sodium hydroxide and protein was estimated according to the method of Lowry et al. [221].
Tubulin polymerization assay:
Tubulin polymerization was determined by GTPase assay, in presence of microtubule reassembly buffer [249]. The reassembly buffer in a total volume of 500 ml contained 5mM MES buffer (pH 6.8), 1mM EGTA, 0.5mM MgCl2 and 50 mm KCL, 100 mg of membrane protein and 20 mg of tubulin (rat brain or plant). After equilibrating the reaction mixture in a shaker water bath (270C) for 10 min, the reaction was initiated by the addition of GTP to 1 mm final concentration. At various time points the reaction was terminated by adding 50% TCA to a final concentration of 10%. Then the tubes were kept for 60 min at 00Cj. The precipitate was sedimented by centrifuging at 5,000 rpm for 10 min. The supernatant was carefully removed and used for estimating phosphate by the method of Fiske and Subba Row [248]. Blanks for GTP, tubulin, and membrane fraction were also run along with the samples. For all combinations zero time controls were also maintained.
TUBULIN SYNTHESIS IN VIVO:
In order to quantify the synthesis of tubulin in response to IBA, hypocotyls segments (20 gm) were treated with IBA (30 mg/ml). After treating the segments with hormone for 30 min, the control and IBA treated segments were incubated 24 hr in 5 ml of nutrient medium containing 25 mCi of [14 C]-chlorella protein hydrolysate. Then the segments were thoroughly washed, homogenized and the membrane fraction isolated and immunoprecipitated as described. The immunoprecipitates were dissociated in equal volumes of SDS-buffer and analyzed on SDS-gels (8%). The gels were then subjected to fluorography.
SDS-Gel electrophoresis:
Soluble proteins (250 mg each), membrane proteins (250 mg each) and immunoprecipitates were analyzed in SDS-gel electrophoresis as described in experimental methods of Chapter I.
Fluorography of polyacrylamde gels:
After electrophoresis of the immunoprecipitates, the slab gels were impregnated with PPO, dried and fluorographed for 15 days at –20oC using India X-ray films (40 ASA) basically according to the procedures described by Bonner and Laskey [246].
RESULTS
Analysis of soluble and membrane proteins obtained from the hypocotyls at different stages of adventitious root initiation:
First of all, the effect of IBA-treatment on the content of soluble and membrane proteins of hypocotyls tissue were examined. The results (Table 12) clearly indicate that there is a striking increase in; the membrane protein content of the IBA treated segments, while there is no significant change in the soluble protein content.
Subsequently, the protein profiles of the soluble and membrane fractions were examined by SDS-gel electrophoresis; the results (Fig. 17) indicate that the soluble protein profiles show certain differences. The conspicuous change is in term of a protein band of molecular weight 1, 05,000 daltons. This protein is detectable at 24hr and 48hr after IBA treatment, but is not seen in the hypocotyls tissue 72 hr after IBA treatment or in untreated tissue.
The protein profiles of the membrane proteins are depicted in Fig. 18. It is clear that a protein band of a molecular weight 55,000 corresponding to the a-subunit of the tubulin dimmer is detectable in IBA treated hypocotyls but not in the control.
Table 12: Effect of IBA on soluble and membrane protein contents.
______________________________________________________________________
Treatment Time Soluble protein Membrane protein Membrane
soluble (mg)
(mg) protein
______________________________________________________________________
Control 0 hr 23.5 2.15 0.09 ± 0.021
24 hr 27.6 2.30 0.08 ± 0.032
48 hr 23.8 1.90 0.08 ± 0.019
72 hr 23.8 1.50 0.06 ± 0.031
IBA 0 hr 23.5 2.15 0.09 ± 0.020
24 hr 24.6 2.90 0.11 ± 0.028
48 hr 25.0 3.20 0.12 ± 0.021
72 hr 32.1 4.60 0.14 ± 0.041
______________________________________________________________________
Soluble and membrane fractions were prepared from the treated and control hypocotyls as explained in Materials and Methods. Protein was estimated by the method of Lowry et al. [221] and the data are presented as a ratio between membrane and soluble proteins. The values represent mean ± S.D obtained from three independent experiments.

Fig. 17: Analysis of soluble proteins by SDS-slab gel electrophoresis.
French bean hypocotyls tissue (30 gm) was treated with IBA (30 mg/ml) for 30 min. After incubation segments were washed and transferred to the fresh basal medium. At 0 hr, 24 hr, 48 hr and 72 hr the tissue was processed for extracting soluble proteins as described in the experimental part. Protein samples (250 mg) were loaded on to each slot, electrophoresed, stained and photographed as described in Materials and methods.
Lane 1 – Standard proteins containing BSA, ovalbumin and cytochrome C-oxidase.
Lanes 2, 3, 4 and 5 are 0hr, 24hr, 48 hr, and 72 hr samples respectively
Fig. 18: Analysis of membrane proteins by SDS-slab gel electrophoresis.
The treatment conditions are as described in Fig 17 Isolation of membrane proteins has been described in Materials and methods.
Lane 1- standard proteins.
Lane 2, 3, 4 and 5-0 hr, 24 hr, 48 hr and 72 hr samples respectively.

Fig. 18: analysis of membrane proteins by SDS-slab gel electrophoresis.
He treatment conditions are described in Fig. 17. Isolation of membrane proteins has been described in materials and methods.
Lane 1- Standard proteins.
Lane 2, 3, 4 and 5; 0hr, 24 hr, 48 hr and 72 hr samples respectively
Identification of IBA-induced membrane protein of Mol. Wt. Of 55,000 daltons as tubulin by SDS-gel electrophoresis
To identify that the new protein band that appeared in IBA treated hypocotyls is tubulin, the tubulin isolated from the rat brain (Fig. 19) was electrophoresed with membrane proteins. Figure 18 depicts that the new protein induced by IBA has the same mobility as purified tubulin. The tubulin protein was also isolated from the plant tissue by the procedure described in materials and methods. The purified preparations show an electrophoretic mobility similar to that of rat brain tubulin in SDS-gel electrophoresis (Fig. 20).
Characterization of tubulin by polymerization assay and immunoprecipitation methods:
Further characterization was carried out by examining the GTP-dependent polymerization of the tubulin fraction. It is known that in reassembly buffer tubulin polymerization is associated with GTP breakdown. The preparation from the plant tissue is found to substitute for rat brain tubulin in the polymerization assay measured in terms of GTPase activity (Table 13) Another interesting feature is that the addition of IBA in vitro strikingly enhances tubulin polymerization as measured in terms of GTPase assay (table 14).
To further confirm the identity of tubulin in IBA-treated hypocotyls, antibodies to rat brain tubulin were raised in rabbits. The IgG fraction prepared from the antiserum shows

Fig 19: SDS-Polyacrylamide gel analysis of purified rat brain tubulin.
Rat brain tubulin was purified as described in experimental procedure. Various fractions obtained during purification were electrophoresed.
Lane 1 – Standard proteins.
Lane 2 – Homogenate.
Lane 3 – 0.2 m NaCl eluates
Lane 4 and 5 – 0.3m NaCl eluates.
Lane 6,7 and 8 –0.4m NaCl eluates.

Fig. 20 SDS-Gel analysis of purified plant tubulin.
Lane 1- Standard proteins containing BSA, Rat brain tubulin, ovalbumin and cytochrome C- oxidase.
Lane 2 and 3 – Rat brain tubulin.
Lane 4 – Plant tubulin.
Table 13: Tubulin polymerization assay reaction.
mMoles of GTP hydrolyzed/mg Membrane proteins
Protein/60 min.
______________________________________________________________________
Plant microsomes 0.65 ± 0.03
Plant Microsomes
+ Plant tubulin 1.50 ± 0.04
Plant Microsomes
+ Rat brain tubulin 1.20 ± 0.03
The reaction was carried in a total volume of 500 ml of reassembly buffer containing 20 mg of plant of rat brain tubulin, 100 mg of membrane protein. The reaction was initiated by the addition of GTP to a final concentration of 1 mm. After 60 min of incubation at 300C the phosphate released by GTP hydrolysis was measured as described in Materials and Methods.
Table 14: Effect of IBA on tubulin polymerization.
______________________________________________________________________
mMole of GTP hydrolyzed/mg membrane
Protein/60 min
_____________________________________
Expriment 1 Expriment 2
______________________________________________________________________
Microsomes 0.66 0.68
Microsomes +
Plants tubulin 1.30 1.18
Microsomes + IBA 0.67 0.63
Microsomes + IBA +
Plant tubulin 3.30 3.60
________________________________________________________________
Tubulin polymerization reaction was carried out as described in Table 13. IBA (10 ng/500 ml) was added to the reaction mixture. Reaction was initiated by the addition of GTP to a final concentration of 1 mm. Phosphate released during GTPase-dependent polymerization was determined as described in Materials and methods. Values given are the mean of two replicates. The results of two independent experiments are given.
A precipitin line with the plant membrane preparations in Ouchterlony double diffusion analysis. The precipitin line fuses with that obtained for the brain tubulin
The quantitative immunoprecipitation curve obtained with the hypocotyls membrane preparation and the tubulin antibody precipitates 3.5 mg of the hypocotyls membrane protein. The immunoprecipitate, when analyzed on SDS-gels, clearly shows the tubulin bands in addition to that of the light and heavy chains of IgG (Fig 23)
Synthesis of tubulin in vivo in response to IBA treatment.
To measure tubulin synthesis in vivo, control as well as IBA treated hypocotyls was labeled with [14C]-chlorella protein hydrolysate for 24 hrs and then the membrane fraction was prepared from the tissue, solubilized and immunoprecipitated with tubulin antibody as described in experimental methods. The immunoprecipitates were washed and subjected to SDS-gel electrophoresis and the gel was analyzed by Fluorography. Figure 24 shows the significant increase in the labeling of tubulin in IBA treated hypocotyls tissue.
Effect of cytochalasin B and colchicines on IBA-induced root formation:
In order to find out, whether microtubules play any role in cell polarity fixation leading to adventitious root initiation

Fig 21. Ouchterlony-Double diffusion analysis of plant membrane proteins with rat brain tubulin antibody.
Ouchterlony planchetes for immunoprecipitation were prepared as described in materials and methods. To the central well purified antibody to the rat brain tubulin was added. The peripheral wells contained membrane protein from (1) control, (2) 24 hr, (3) 48 hr and (4) 72 hr IBA-treated segments. Wells 5 and 6 contained rat brain tubulin and BSA respectively.

Fig 22. Titration of rat brain tubulin antibody with plant membrane proteins.
The procedure for immunoprecipitation has been described in materials and methods. Increasing amount of solublilized membrane proteins were added to 550 mg of IgG fraction in a total volume of 0.5ml. The mixture was incubated at 40C overnight. Separate antigen and antibody Blanks were maintained. The immunoprecipitates were washed and used for protein estimation as described in the text. The values were the mean of two replicates.

Fig. 23: SDS-PAGE analysis of the immunoprecipitated plant membrane tubulin.
Hypocotyl tissue was treated with IBA for 30 min and then untreated as well as IBA-treated hypocotyls segments (20 gms each) were incubated with 5 ml of basal medium containing 25 mCi of [14C]-Chlorella protein hydrolysate for 24 hr. The segments were thoroughly washed and the membrane protein was immunoprecipitated with the rat brain tubulin antibody. The immunoprecipitates were analyzed on SDS-slab gels and stained
With Coomassie Blue.
Lane A – Standard proteins containing BSA, RAT brain tubulin, ovalbumin and cytochrome C- oxidase.
Lane 2 – Immunoprecipitates from control segments.
Lane 3 – Immunoprecipitates from IBA treated segments.
Fig. 24: Fluorograph of Immuniprecipitated plant tubulins.
The gel depicted in Fig. 23 was fluorographed.
Lane A – Tubulin from control tissue.
Lane B – Tubulin from IBA treated tissue.

Fig. 24: Fluorograph of Immuniprecipitated plant tubulins.
The Gel depicted in Fig.23 was fluorographed.
Lane A- Tubulin from control tissue.
Lane B- Tubulin from IBA treated tissue.
The effects of colchicines and cytochalasin B on IBA mediated root initiation in the hypocotyls tissue were examined. The tissues were exposed to cytochalasin E or colchicines for 6 hr. The results (Figs. 25 and 26) indicate that the two drugs effects are clearly seen if the IBA-treated tissues receive the colchicines or cytochalasin B treatments at any time before 36 hr. Treatment of the IBA exposed tissues at any time periods after 36 hr has no effect on root production.
DISCUSSION
The results obtained in the present study clearly establish that the protein with 55,000 molecular weight synthesized within the hypocotyls tissue in response to IBA treatment is tubulin. It has been possible to isolate a protein fraction from the hypocotyls tissue exhibiting the properties of tubulin, by the procedures employed to purify brain tubulin. The plant preparations show a molecular weight similar to that of brain tubulin in SDS-gels. It can also substitute for brain tubulin in polymerization assay. The antibody to brain tubulin shows reaction with the plant protein preparations.
It has also been possible to demonstrate the IBA treatment results in a striking increase in the rate of tubulin

Fig. 25: Effect of cytochalasin-B on IBA-induced adventitious root formation.
Hypocotyl segments (8 cm length) were first treated with IBA (30 mg/ml) for 30 min. Then the segments were washed and transferred to fresh basal medium. At different time points segments were transferred to flasks containing basal medium with cytochalasin-B (10 mg/ml) and incubated for 6 hr. After washing the segments in running water for 30min, they were transferred to fresh medium and the medium was changed frequently. The roots were counted at the end of 72 hr. The average number of roots obtained from 10 segments was given with the standard deviations

Fig. 26: Effect of colchicine on IBA-induced root initiation.
IBA treated hypocotyls segments were treated with Colchicine (0.24%) as described in the Fig. 25. The average number of roots obtained from 10 segments was given with standard deviations.
The demonstration in the previous chapter that the 55,000 molecular weight protein is conspicuous among the protein products synthesized in the wheat germ cell-free system programmed by mRNA from IBA-treated hypocotyls tissue, indicates that perhaps the hormone increases the tubulin mRNA synthesis.
Another interesting feature is that IBA addition in vitro enhances tubulin polymerization strikingly as measured in terms of GTPase assay (Table 14). These results indicate that the hormone, in addition to causing the synthesis of tubulin and its mRNA may also be involved directly in the tubulin polymerization process and thus influence the formation of cytoskeletal structures. In this context it is of interest to point out that nerve growth factor is also capable of this direct interaction with tubulin [247]. This direct interaction may be involved in the process of growth and differentiation. For example, one of the early effects of auxin treatment is cell elongation and the direct rapid interaction of the hormone with tubulin and MAPS may be involved in this process. Subsequent induction of tubulin synthesis coupled with the direct interaction of the hormone with tubulin and its associated proteins catalyzes its polymerization may also be involved in cell commitment to differentiation.
The ability of cytochalasin B and colchicines to block IBA-mediated root indicates the involvement of microtubules in polarity fixation leading to new root formation. The cells in the pericycle region above the split exarch xylem strands appear to be irreversibly committed to root formation between 30-36 hr after IBA treatment.
The transient appearance of 1, 05,000 molecular weight proteins in the cytosol of IBA-treated tissues further testifies as to the ordered programming of protein synthesis under conditions of hormone treatment. While the involvement of such proteins in differentiation process is not clear at the moment, it is known that a variety of auxiliary proteins such as MAPs and TAU [17, 18, and 247] are involved in tubulin polymerization, thereby influencing the formation of the cytoskeletal structures, orientation and organogenesis.
SUMMARY
1. There is a striking increase in the membrane protein content in the IBA-treated hypocotyls segments.
2. A high molecular weight soluble protein (1,05,000 daltons) is conspicuously found at 24 hr and 48 hr after IBA treatment, but is not seen in the hypocotyls tissue 72 hr after IBA treatment or in control tissue.
3. A membrane protein band of molecular weight 55,000 daltons corresponding to the a-subunit of the tubulin dimmer is detectable in IBA treated hypocotyls but not in control tissue.
4. Tubulin has been isolated and purified from rat brain and hypocotyls tissue.
5. Based on SDS-gel electrophoresis, tubulin polymerization assay in vitro and immunoprecipitation methods, the 55,000 molecular weight membrane protein induced by IBA has been identified and characterized as tubulin.
6. Tubulin synthesis in vivo in response to IBA treatment has been determined by immunoprecipitation and flurographic methods.
Adventitious root formation is inhibited, if the hypocotyls segments are treated with cytochalasin B and colchicines within 36 hr of IBA treatment, suggesting the involvement of microtubules and microfilaments in new root formation.