MORPHOGENESIS- A Review
G. R. KANTHARAJ* and G. PADMANABHAN**
* Dept. of Botany, The National Collage, Bangalore – 560 004, India. (Correspondence address)
** Dept. of Biochemistry, Indian Institute of Science, Bangalore - 560 012, India
Morphogenesis involves growth and differentiation. Growth generally refers to the enlargement of the cells or the production of a group of cells of similar kind. In contrast to growth, differentiation involves the development of a cell or an organ which is structurally and functionally different from the cell it originates. In the development of a plant both growth and differentiation, go hand in hand. Plants like animals start their development from an unicellular structure, which by a series of pre-determined cell divisions produces a variety of cell structures and organs. Except for a few cell types such as sieve tubes among phloem elements, almost all living cells of all kinds are totipotent and if proper nutritional conditions are given they are capable of giving rise to a complete plant. This developmental potential is inherent in their genetic make-up. In recent years, however, it is becoming clear that the differential expression of the genetic potential is controlled by plant growth substances that are produced in the system itself. It is their interaction with the genetic apparatus which results in the full expression of the phenotype of the plant.
PLANT GROWTH REGULATING SUBSTANCES: HISTORICAL ACCOUNT:
The existence of a plant growth substance was first perceived by Charles Darwin [1], who in his own elegant way, described how plants perceive the stimulus of light and respond to phototropic growth. Since then, botanists have made great strides in unraveling the mystery of the diffusible substance, which is responsible for growth. Kogl and Haagen Smit isolated and identified the growth promoting substance and coined the name “hetero auxin” or as it is known today, indole-3-ylacetic acid (IAA) [2,3]. This gave a great impetus to the plant scientists all over the world, and the search culminated in a series of discoveries of plant growth substances, such as Gibberellin B (GAB) by Yabuta and Sumiki (1938), Cytokinin by Miller and Skoog (1955), and Abscisic acid (ABA) by Robinson and Wareing (1964). Simultaneously a host of synthetic growth promoting as well as growth inhibiting substances were also made available to plant scientists. This led to intensive investigations on the effect of the various plant hormones on plants. Various hormones, elicited a wide array of responses in different kinds of tissues. It was soon realized that the physiological response to any given growth substance depends, in the first instance, on the type of cell receiving the stimulus. For example, a developing leaf cell, in a barley plant, will respond to GA3 by elongating, whereas an aleurone cell, from the same plant, will respond by producing a-amylase. The specificity of response is built into the cell by its previous developmental programme. It is curious to note that the highly specific animal hormones affecting specific cells have no counterpart in plants. In general, it can be stated that the auxin is known as growth promoting substance, but the same substance is also capable of eliciting responses such as, apical dominance, root initiation, prevention of leaf abscission, fruit setting and so on. While Gibberellic acid is known to be effective in promoting growth, induction of a-amylase, bolting and flowering. Cytokinin is considered s the hormone that controls cytokinesis, a part of cell division. Abscisic acid is known to act as a growth inhibitor, controlling processes such as bud dormancy and seed dormancy. Ethylene, recognized as a hormone recently, shows bizarre effects. In the development of the plant body, it is the interaction between the various hormones that controls and regulates the process of morphogenesis.
HORMONAL INTERPLAY
Although Haberlandt (1913) considered the tissue and organ culture in sterile conditions as a theoretical possibility, it was realized as a practical proposition for research by two independent workers, Kottle (1992) in Germany and Robbins (1922) in America. The advent of tissue culture techniques opened up a new vista in the studies of hormonal interactions in morphogenesis.
When plant growth substances are supplied to plant tissues exogenously, each of them elicits unique responses. However, many responses are overlapping. But if two or more different hormones are supplied in known concentrations, in many cases synergistic or antagonistic responses are exhibited and this effect is referred to as hormonal interaction.
Studies on tobacco pith callus tissue have revealed that the relative concentrations of hormones control the expression of callus either into roots or shoots. Low, high and intermediate ratios between auxin and cytokinin induce roots, shoots and callus respectively [4]. Auxin-cytokinin-induced shot formation in the callus is inhibited by GA3; however GA3 enhances the growth of the callus. The inhibitory effect of GA3 can be alleviated by the addition of ABA, which by itself is a weak inhibitor of callus growth [ 5,6 ].
The form of shoots developed from callus is dependent on GA3-cytokinin ratio. The higher ratios induce the formation of tall, spindly shoots with narrow leaves, whereas lower ratios produce dwarf shoots with rounded leaves [7].
Specific hormones are found to influence the relative levels of other hormones and this is very well substantiated in the culture system, where auxin-cytokinin induced callus growth is reduced by certain culture conditions, which also inhibit the biosynthesis of GA [6]. Thus, the interactions of hormones appear to be very complex.
This complexity is further compounded by the recent observations, in which, it has been shown that each hormone can influence the biosynthesis, degradation, conjugation or transport of one or the other hormones. The endogenous level of IAA in a plant tissue is enhanced by the exogenous application of GA or Kinetin. But, ethylene, which is known to be induced by excess auxin, reduces the level of auxin itself. Again, auxin levels can be increased by the addition of cytokinin. This complex relationship appears to be very fascinating, because of the findings in which cytokinins are known to bring down the levels of ABA, through the increased biosynthesis of GA. But ethylene promotes the synthesis of ABA, which in turn enhances the levels of ethylene. The close relationship between ethylene and auxin provides an excellent feed back control mechanism. Letham et al. (1978) have illustrated the complex interaction between plant growth substances as indicated in the diagram (Fig. 1) [8]. Further investigations into hormonal interactions at molecular level, will have far reaching impact in understanding morphogenesis at the molecular and sub cellular levels.
ULTRASTRUCTURAL CHANGES DURING DIFFERENTATION: MICROTUBULES, MICROFILAMENTS IN CELL POLARITY AND ORGANOGENESIS:
The temporal and quantitative changes in the regulation of gene activity forms the basis for the ultra structural changes determining the process of cell development and
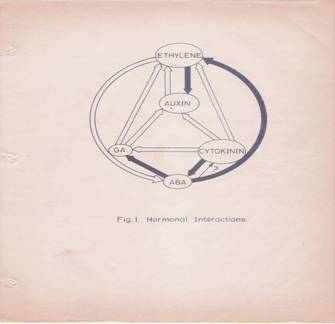
Fig. 1: Hormonal Interactions.
![]()
![]() Fig.1: Hormonal interactions which affect
hormonal levels in plant tissues. Open arrow from A B indicates hormone A
causes increase in the levels of B. Closed arrow from A B indicates that
hormone A decreases the levels of B. differentiation.
Fig.1: Hormonal interactions which affect
hormonal levels in plant tissues. Open arrow from A B indicates hormone A
causes increase in the levels of B. Closed arrow from A B indicates that
hormone A decreases the levels of B. differentiation.
The molecular events leading to such ultra structural organization are not very clear. For example, it is not clear how and why a fertilized egg in the embryo sac, always undergoes a polarized division in such a way, that one of the two cells, equal or unequal, develops into radicle and the other into plumule. This polar behavior of the cells at the earliest part of their development is the function of intracellular compartmentalization, regulated and governed by certain intrinsic interactions of nucleo-cytoplasmic components, whose expression in turn is controlled by the inherent genome.
Polar growth like cap (reproductive organ) formation in Acetabularia, rhizoidal development in Fucus, hold-fast development in Cladophora are some of the best studied systems in the plant kingdom. In the case of zygote development in Fucus, if the spherical zygote is subjected to unidirectional light treatment, the rhizoid develops at the shaded side of the cell. If such a programmed zygote is subjected to electron microscopic investigation, it is observed that in the region of rhizoid formation, large number of osmophilic bodies and undefined extra cellular materials accumulate. The perinuclear region has shown highly polarized finger shaped projections towards the site of rhizoid initiation. Along with these charges, heavy concentration of mitochondria, ribosomes and dense fibrillar vesicles are also found. These structural changes represent an expression of a fixed axis for rhizoidal growth [9]. The time taken for such polarity fixation has been determined to be 10-16 hrs. During this period, if the cells are treated with cycloheximide (CHI) for 9-15 hrs after fertilization, they still show a fixed polar axis. However, if cytochalasin-B, which is known to disrupt the microfilaments, is present during polarity fixation, the polar axis remains labile and axis establishment is prevented [10].
The role of cytochalasin-B and colchicines, in lectin Con-A induced “cap” formation in animal cells, has been reviewed by Edelman (1976) and Nicholson (1976) [11,12]. The authors have explained how the microtubules and microfilaments control the mobility, stabilization and topographical distribution of cell membrane and surface components during lectin induced “cap” formation. The cross linking of membrane receptors embedded in the plasma membrane, to a variety of fluorescent labeled polyvalent ligands such as antibodies or lectins lead to form patches and clusters which ultimately culminates in the “cap” formation at one end of the cell. Similar to “cap” formation in animal cells, “cortical clearing” in the region of rhizoidal emergence is observed in zygotes of the plant Pelvetia fastigata [13]. In the same system, Peng et al. (1976) have shown the deposition of new membrane patches (30 um) covering the rhizoidal region at the time of cortical clearing [14]. These studies clearly suggest that microtubules and microfilaments play a significant role in cell polarity, which is a prerequisite for organogenesis. Ever since, the discovery of micro tubular components in few plant cells by Lederbetter and Porter (1963) [15] the presence of such structures, in most of the plants, has been established.
Basic components of the microtubules are not known to be tubulin monomers, which exist in the form of heterodimers, consisting of a and b subunits, of molecular weight 110,000- 120,000 daltons [16]. Several proteins are associated with microtubules, of which the proteins known as TAU and microtubule associated proteins (MAPS) appear to be important, for they take part in assembly and disassembly of microtubules [17,18,19]. In addition to the above components, the presence of nucleating centers or organizing centers within the cells, account for an ordered or controlled assembly of microtubules [20,21]. In a developing system, microtubule assembly is under close spatial and temporal regulation. The nucleating centers function in controlling initiation, orientation, directionality and patternization of micro tubular polymerization. Recently attempts have been made to isolate microtubules and study their organization in plant tubulin [22,23]. The cross reaction of antibodies, raised against porcine brain tubulin, with the plant protein, strongly suggests the similarity of plant tubulin with animal tubulin, at least at the antigenic level. Considerable attention has been given to relate the orientation of microtubules during plant hormone induced growth. Hormone induced growth in Vigna angularis, hypocotyl of Lactuca sativa and coleoptile segments of Triticum vulgare, is inhibited by colchicine, urea and vineblastin sulfate [24-26] and this inhibitory effect is attributed to micro tubular disruption [27]. Similarly, the inhibition of protoplasmic streaming in Avena, and growth inhibition in Zea mays root by cytochalasin-B are again attributed to the role of microfilaments and microtubules [28,29]. Electron microscopic studies on the deposition of cellulose in cell walls during secondary growth and movement of secretory vesicles are shown to be directed by microtubules [30]. However, the available information on plant hormone induced changes in the micro tubular assembly, synthesis and their role in morphogenesis is scanty.
PLANT HORMONES AND NUCLEIC ACID METABOLISM
Plant growth regulating substances, very often, exhibit dramatic effects on growth and development in plants. The molecular mechanisms which control such processes involve changes in nucleic acid and protein metabolism. DNA replication, transcription and translation are the three major molecular events involved in growth and differentiation. An understanding, as to how plant hormones control such molecular mechanisms, as to how plant hormones control such molecular mechanisms, is very pertinent to interpret morphogenesis at the molecular level. In spite of the enormity and complexity of the problem involved, considerable Progress has been made in this field.
DNA METABOLISM
A majority of investigations reveal that hormone induced cell elongation does not require DNA synthesis [31,32]. However, Wardell (1975) has reported that auxin, in flowering tobacco plants, enhances DNA synthesis within an hour of treatment. On the contrary, in pea root cortical cell cultures [33], Libbenga and Torrey (1973) have shown that enhanced DNA synthesis is possible, only in the presence of auxin and kinetin together, but not with auxin alone [33a].
In long term experiments, most of the growth promoting substances enhances DNA synthesis which is always accompanied by cell division [34]. In cucumber hypocotyls, GA3 – promoted growth is fluoro-deoxyuridine (FUDR) sensitive. This increase in DNA synthesis, in response to GA3 is due to the increased chloroplast proliferation, which is prevented by chloramphenicol. The above experiments suggest that GA3 has some preferential effect on chloroplast biogenesis [35, 36].
On the other hand, hormones such as ethylene and abscisic acid inhibit DNA synthesis. This correlates well with their growth inhibitory effects in vivo [37-39]. However, it is not clear whether this effect is direct or indirect.
Although, cytokinin is known to induce cell divisions, it does not affect DNA synthesis by itself as a prelude to cytokinesis. However, it promotes DNA synthesis in the presence of auxin [40]. Hence, it is suggested that auxin has a permissive role in DNA synthesis, while kinetin stimulates it. This is further exemplified in an experiment, where hormone depleted and aged tobacco pith explants, could be induced to undergo mitosis without the accompanying DNA replication by the addition of cytokinin. This suggests that cytokinin perhaps regulates cell division, by controlling the synthesis of specific proteins for mitosis rather than acting directly on DNA replication [41,42].
DNA SEQUENCE COMPLEXITY
In higher plants, it is estimated that the amount of DNA present per cell ranges from 1pg –100 pg and this amount of DNA codes for more that 106 different proteins. The rough estimates indicate that there are 50,000 or more structural genes which are expressed at one time or the other during the life of the plant [43]. The number of structural genes operating in different plant structures like root, stem, leaves and flowers may vary. On the basis of hybridization studies between polysomal RNA and 3H-labelled single copy DNA, it has been estimated that there are about 2700 diverse structural gene transcripts in the tobacco leaf cells [44]. DNA, in higher plants, consists of three base sequence classes i.e., highly repeated, moderately repeated and unique. Unique sequences are believed to code for mRNA sequences [45]. These sequences can be detected, characterized and quantified by studying the reassociation kinetics of denaturated and sheared DAN. Highly repeated sequences reassociate rapidly at low cot values (cot = concentration of DNA in mole nucleotide per liter X time in seconds) and unique sequences reassociate slowly at high cot values [45]. Studies on changes in DNA sequence complexities in response to phytohormones in plants are few. Nevertheless, reannealing studies of DNA isolated from the control and auxin treated artichoke tubers; suggest that auxin causes an apparent loss in the rapidly reannealing fraction of DAN, but leads to some dramatic changes in the cot values for other fractions [46]. On the contrary, GA3 has no effect on the DNA isolated from the roots of Cucumber but DNA from the treated shoots and leaves of the same plants, exhibit a nine-fold increase in the intermediate reassociation fractions. According to Britten and Davidson (1969) such sequences may have a regulatory role [47]. In cymbidium protocorm tissue, while auxin causes an increase in AT-rich DNA, GA3 promotes an increase in GC-rich DNA [48]. Similar studies have been carried out in various plants such as wheat embryos, pea epicotyls etc. [49]
However, the above studies do not indicate whether, phytohormone induced changes in DNA population is due to primary or secondary effects of the hormones.
PHYTOHORMONES AND TRANSCRIPTION
Protein synthesis is a crucial event in growth and differentiation, and therefore the effect of phytohormones on transcription and processing of different kinds of RNA would determine the process of morphogenesis.
AUXIN-MEDIATOR/ACCEPTOR PROTEINS AND RNA SYNTHESIS
The mechanistic aspects of auxin function appear to be very complex. Previous investigations have suggested that there may be one or two cellular sites at which auxin may act and elicit its responses on growth and differentiation. One such site is found to be located in the plasma-membrane and the other may be a non-plasma membrane site located either in the cytoplasm or nucleus.
Auxin-mediated early cell enlargement is known to function through the activation of membrane bound factors [50-52]. The early growth is insensitive to both cycloheximide and actinomycin D; hence neither RNA nor protein synthesis is required for this function. However, the second phase of growth requires both RNA and protein synthesis. The auxin-induced mechanism of early growth is speculated as due to the cell-wall loosening effect caused by the secreted protons, which are pumped out by the auxin-activated membrane bound factors [53]. The existence of soluble auxin-binding proteins of molecular weight 3,15,000 and 10,000 daltons have been reported in dwarf bean seedlings and soybean cotyledons respectively [54,55]. It is not clear, whether these auxin binding proteins have any role in transcription. Hardin et al. (1972) [56-58] have demonstrated that in soybean hypocotyls and onion stems, auxin treatment results in the release of a plasma membrane factor into the cytosol, which stimulates a-amanitin sensitive RNA polymerase activity. However Mathyse and Philips (1969) [59] have shown the existence of auxin-receptor complex in the nuclei and this factor is responsible for the enhanced RNA synthesis. Auxin-receptor protein has been isolated from coconut nuclei and shown to have a molecular weight of 10,000 daltons. It binds to the auxin non-covalently at a molar ratio of 2:1. In the presence of transcribing system, consisting of a-amanitin sensitive RNA polymerase (presumably RNA polymerase II), native DNA, and an initiation factor, the auxin binding factors stimulate RNA synthesis 2-3 fold, only in the presence of auxin [60, 61]. Although, these factors are not well characterized, this constitutes a good evidence for the existence of auxin-receptor proteins.
Using changes in melting point profiles and bathochromic shifts in DNA and chromatin, Fellenberg (1971) and Bamberger (1971) have claimed that auxin interacts with DNA and chromatin directly, thereby facilitating transcription [62,63]. However, these observations turned out to be due to changes in the PH of the medium, rather than to the direct effects [64].
While the studies on auxin-histone interactions have not yielded any significant information, non-histone proteins are now considered to be the most likely candidates influencing specific gene transcription. Non-histone proteins are heterogeneous and include a number of enzymes and structural proteins. They are species-specific and tissue-specific. Their phosphorylation pattern changes with the different states of gene activity [65, 66]. Such an involvement is very well exemplified in the studies involving progesterone and estrogen receptor complexes with the chromatin of target cells in different animal systems [67].
In plants, the existence of zone-specific non-histone proteins is known in soybean hypocotyls [68]. Recently, 2, 4-dichlorophenoxy acetic acid (2, 4-D) induced changes in the phosphorylation of nuclear proteins has been shown in soybean, but the correlation between auxin induced RNA synthesis and the phosphorylation pattern ahs not been established unequivocally [69].
Tissere et al. (1975) have isolated factors from the acidic fraction of the chromatin proteins from the lentil roots, referred to asa, b, g and d factors. On auxin treatment, thea, b and d factors remain unchanged but the g factor increases two-fold. While the g factor regulates RNA polymerase I, d affects polymerase I and II. Hence these factors are known to regulate the activity of RNA polymerases without enhancing the synthesis of the enzyme [70].
Generally, auxins induce RNA synthesis. Silber and Skoog (1953) for the first time correlated auxin-induced cell enlargement with enhanced RNA synthesis [71]. Since then, considerable work has been carried out on the auxin induced changes in the levels of various RNA species.
Auxin has been reported to preferentially increase rRNA content in pea epicotyl and soybean hypocotyls [72]. But, it has also been shown that the enhancement in RNA synthesis occurs well after auxin has effected growth. It has been found that 5-fluorouracil [5-FU] does not prevent cell enlargement in soybean hypocotyls and artichoke tuber, but auxin induced growth in oat coleoptile and soybean hypocotyls is inhibited by actinomycin D. The above observations suggest that auxin induced growth in the earlier stages does not require RNA synthesis, but requires the synthesis of messenger RNA synthesis later [73, 74].
Investigations, using polysome content as a measure of quantification of mRNA synthesis have shown that auxin induced polysome levels are actinomycin D sensitive, but 5-FU insensitive. This indicates that the synthesis of new ribosomes is not necessary for the increase in the levels of polysome [75-78]. Verma et al. (1975) using in vitro cell free protein synthesizing system and cellulase specific antibody, have shown that the auxin induces the synthesis of cellulase-specific RNA [79]. However, increase in other m RNA populations is not ruled out.
It is not clear, whether, the auxin induced messenger RNA is synthesized as a precursor heterogenous RNA (hnRNA) in plants. In short-term labeling experiments (45 minutes), auxin enhances the labeled precursor incorporation, preferentially into the nuclear fraction. Long term labeling experiments; however indicate that incorporation into both nuclear and cytosolic fractions is enhanced [80]. Recent studies with Petroselium sativum (parsley), have demonstrated that most of the poly (A)-RNA is synthesized as hnRNA. This is established by DNA-RNA hybridization-kinetic studies [81].
Attempts have also been made to use Escherichia coli RNA polymerase core enzyme, to measure transcription of DNA/chromatin, as an index of differential gene expression in chromatin, isolated from auxin treated soybean seedlings has been found to code for synthesis of many fractions of RNA species, of which the synthesis of TB-RNA, several fractions of heterodiesperse RNA and 4S-5S RNA are much more than the control. This chromatin preparation enhances labeled amino acid incorporation into proteins by 2-3 folds in coupled transcription-translation assays [82, 83]. Similar studies involving the plant tissues have clearly indicated that auxin can influence polymerase activity, as well as template activity.
GA-BINDING/MEDIATOR PROTEINS AND RNA SYNTHESIS
The presence of GA3 – binding protein in pea stems, epicotyls and other plants has been reported [84, 85]. The approximate molecular weight of the GA3 – binding proteins, extracted from dwarf epicotyls of pea plant, has been determined to be 5, 00,000 and 60,000. Both the fractions bind GA3 noncovalently and easily exchange with non-labeled GA3.
Not much is known about the effects of GA3 binding proteins on transcriptional events. Isolated pea nuclei do not respond to exogenously added GA3 but, if the nuclei are isolated after GA3 treatment, transcription is enhanced [86]. Attempts to localize GA3 – binding sites, in cells by autoradiography or by cell fractionation, have yielded limited results [87]. GA7 has been shown to interact with DNA containing higher AT than GC content. It is also reported that GA7 interacts With DNA isolated from a number of plant sources, as well as some phages, but it does not interact with either animal or bacterial DNA. This specificity of GA7 interaction has not been explained [88]. In the presence of DNA Ligase, GA7 causes the formation of covalently linked loops in cucumber DNA, but cytokinins and auxins do not have any effect [88a]. Recently, it has been reported that at low concentrations of ethidium bromide, GA4, GA7, or GA3, show synergistic effects on elongation in cucumber hypocotyls, perhaps by exposing binding sites in DNA and thus making gibberellic acid more effective [89].
Studies on the effect of GA3 on transcription of either isolated chromatin or isolated nuclei from pear or cucumber show, that the increase in RAN synthesis is due to increased RNA polymerase, as well as due to the increased template availability [90]. A wide variety of tissues such as barley leaf segments, clover seedlings, lentil epicotyls, potato buds, cucumber hypocotyls and sugar beet have been used to study GA3’s effect on RNA synthesis. A significant amount of information on the effect of GA3 on RNA synthesis comes from the studies with barley aleurone cells. GA3 causes a selective increase in m RNA for a-amylase, which on release to the endosperm facilitates the mobilization of carbohydrate source for the growing embryo. If actinomycin-D is added along with GA3, the increase in a-amylase synthesis is inhibited [91]. The labeling of poly (A) – RNA, specific for a-amylase increases after GA3 treatment with a lag period of 3-4 hrs. This correlates well with the lag period of a-amylase synthesis observed in vivo [92]. However, Higgins et al. (1976) and Carlson (1977) have suggested that GA3 activates the preexisting a-amylase m RNA by post transcriptional process into an active translatable form of m RNA [93, 94], but Rodaway et al. (1978) [95] have considered this possibility as very unlikely similar effects of GA3 on poly (A)-RNA synthesis have been established in etiolated maize seedlings [96], but their translational products have not been characterized. It is now known GA activates alpha amylase gene during germination cereal grains
CYTOKININ-RECEPTOR PROTEIN AND RNA SYNTHESIS
The presence of cytokinin-receptor protein has been indicated by the work of Mathyse et al. (1969) [97]. The cytokinin-protein complex enhances RNA synthesis in vitro in presence of pea bud chromatin or DNA as template and E. coli or calf thymus, kinetin-receptor does not stimulate RNA synthesis. Similar studies using nuclei-rich preparations from soybean, tobacco callus and pea buds, have shown that the cytokinins enhance [14 C] –ATP incorporation into TCA perceptible and KOH digestible RNA fractions by 40-100% within 5-10 minutes after treatment. To the phytohormone-starved tobacco pith callus, the addition of cytokinin alone does not cause any major charges in RNA metabolism, but cytokinin with auxin treatment enhances [32 p] – orthophosphate incorporation into polysomal RNA within 3 hrs of treatment [98, 99]. On the contrary, in the hypocotyls of soybean, even at early stages after treatment, the auxin induced incorporation of [14 C]–urudine into RNA is inhibited by kinetin. After 6 hrs of incubation with cytokinin and auxin together, the inhibition of labeled precursor incorporation into rRNA and tRNA is 90% and TB-RNA 30-50%. However, the labeling into D-RNA is not affected [100-103].
Cytokinin-mediated increase in the specific isoacceptor leucyl-tRNA species has been detected in cotyledons and hypocotyls of beans [104, 105]. This could be the result of and effect of direct synthesis or on post-transcriptional modifications. In general, it seems that the occurrence of cytokinins in tRNA is not longer viewed as necessary for eliciting the cytokinin response [106-108].
ETHYLENE-RECEPTOR AND RNA METABOLISM
Although no coherent information is available regarding ethylene binding to receptor proteins, transcriptional studies with isolated chromatin from ethylene treated tissue, show qualitative changes in the RNA species synthesized. There is a change in the nearest neighbor frequency of the RNA synthesized, thus indicating transcription of new template in response to ethylene [37].
Generally ethylene is known to induce abscission and fruit ripening in plants. During abscission, ethylene enhances the synthesis of cellulase and pectinase, which are responsible for the breakdown of the cell wall and pectin [109, 110]. In cotton, Coleus and bean plants, ethylene induced abscission can be prevented by actinomycin D. Furthermore, using 5-FU as an inhibitor of rRNA synthesis, investigations have demonstrated that the ethylene mediated abscission involves induction of specific mRNA, but the products have not yet characterized.
Similar results have been obtained in the ethylene induced ripening of Pyrus mallus fruits [111]. In ripening tomato fruits, ethylene brings about changes in the relative amounts of iso-accepting species of leucyl-, lysyl-, methionyl- and tyrosinyl-tRNAs.
ABA-RECEPTOR AND RNA SYNTHESIS
ABA affects a number of physiological processes, such as dormancy, leaf senescence, abscission, seed germination and flowering, and also antagonizes the effects of other plant hormones, in a number of plant tissues. It is not clear, whether such physiological responses to ABA are brought about through mediator molecules. However, various reports indicate that the effect of ABA is on RNA – turnover and translational process.
ABA has been shown to inhabit the transcriptional capacity of isolated chromatin [112]. This inhibitory effect is ascribed to the effect of ABA on polymerase activity [113,114]. In Fraxinus embryos [115], ABA inhibits labeled precursor incorporation into RNA, and reduces polysome formation, but protein synthesis is not affected. The above results have been interpreted to mean that ABA prevents the synthesis of mRNA and other RNA species, thereby affecting growth.
ABA, in pear embryo and lentil roots, causes a preferential increase in UMP rich RNA and a substantial decrease in GMP, but there is not change in AMP, CMP contents of RNA (115 a). This observation is significant in view of the recent observation, that translation control RNA (tcRNA), which controls translation of certain mRNAs has very high (50%) content of UMP nucleotides [116]. Additional support to the above observation comes from the findings that ABA treated germinating seedlings contain UMP rich (40-50%) RNA species [117]. It is not clear whether this high level of UMP rich RNA is due to new synthesis or preferential degradation of the other RNA species.
ABA also inhibits GA3 – promoted a-amylase synthesis, in barley aleurone cells. They act antagonistic to each other. Some investigators have shown that ABA inhibits [14 C] – uridine incorporation into poly-disperse RNA [92, 118]. There are also reports that ABA does not inhibit the synthesis of any species of RNA [119]. In this context, the observation about certain short chain fatty acids (of C5 and C9 length) at a concentration of 10-4 M, inhibiting GA3 – induced amylolysis in barley endosperm is interesting. Such short chain nonanoic fatty acids have been detected in many germinating seeds such as oat and fenugreek [120].
PHYTOHORMONES AND POST – TRANSCRIPTIONAL EVENTS
Perichromatin and Precursor RNAs:
Most of the RNA species, transcribed within the nucleus, are in the form of larger molecules and these are processed and transported to the cytoplasm across the nuclear membrane pore complex [121]. The existence of extra chromosomal and extra nuclear ribonucleoprotein structures called perichromatin granules (presumably precursors of mRNAs) in rat liver nuclei and plant cells, has been reported as early as 1969 [122]. The transport of such particles across the membrane pore complex has been speculated. Active movement of such chromatoid bodies (containing RNA) across the pore complex, during rat spermatogenesis is very well documented [123]. In plants, synthesis of large precursor RNAs (hnRNA) and their processing into the mature forms have been reported [43,124,125] and well established.
POLY (A) AND “CAP”
During the processing of hnRNA, additional structures such as the “cap” at the 5’ end poly (A) chain at the 3’ end are added to the mRNA. Addition of poly(A) segments of 50-200 nucleotides, at the 3’ end of the hnRNA or premRNA is catalyzed by poly(A)A polymerase associated factors [126]. The existence of such polyadenylated hnRNA and pre mRNA has been demonstrated in the nuclei of plant cells [93,127,125]. However, not all mRNAs contain such additional structures; RuDP carboxylase in chloroplasts is one such example [128].
Capping at the 5’ end of the mRNA involves the addition of 7-methylguanosine, which is linked by a 5’-5’triphosphate bridge to a the first nucleotide of pre mRNA, which can again be methylated at 2’o position [129,130]. The “cap” structure is implicated in influencing the processing of pre-mRNA and initiation and efficiency of translation [131, 132]. However, “cap” is not required for the translation of all mRNAs [133]. Recent reports suggest that the prokaryotic mRNAs, which are “capped” in vitro, translate very efficiently in cell-free eukaryotic protein synthesizing system [134]. Sonenberg et al. (1979) have isolated a “cap” binding protein whose molecular weight is 24,000 daltons. This protein stimulates translation of capped mRNAs in an in vitro system derived from HeLa cell extracts, under conditions that do not increase translation of non-capped mRNAs from satellite necrosis virus [134a]. In plants, there are not many reports about “cap” structures in mRNAs. However, addition of 7-methyl guanosine 5’-monophosphate (m 7G5P) to the French bean seed mRNA – programmed cell free protein synthesizing system derived from wheat germ, inhibits [35 S]- methionine incorporation into proteins by 90%, thereby suggesting that the mRNA isolated from French bean cotyledons are “capped” [135]. The role of plant growth substances in the “capping” process is not clear.
The non-coding sequences found between the first AUG codon and the “cap” at the 5’ end and the last terminator codon and the first adenine nucleotide of the poly (A) segment at the 3’ end are suggested to play an important role in initiation and termination of protein synthesis [136]. Such information is not available with plant mRNAs.
INFORMOSOMES
The messenger RNA is transported from the nucleus to cytoplasm as m RNA protein complex (m RNP). These m RNP particles may release the m RNA for polysome formation or the m RNPs may remain in an inactive form. The latter particles are referred to as informosomes. Such informosomes are found in a number of embryonic tissues [93,137,138] and other non-embryonic structures such as stems, tubers, etiolated leaves, dry mosses, spores of ferns, Acetabularia and Dictyostelium [14,103,139,140,142,143,144]. Such inactive m RNPs have also been reported in animal cells [145-147].
The m RNPs that are found in most of the organisms have poly(A) tail at 3’ end [148]. The presence of such m RNPs ensure the preservation of m RNAs during periods of dormancy and certain adverse environmental conditions such as drought, lack of optimal photoperiodic stimulus and lack of light of actinic wavelength.
ACTIVATION AND INACTIVATION OF m RNAs
The activation of preexisting dormant m RNPs to external stimuli has been reported in a variety of systems such as germinating seeds [138,159] rejuvenating moss[141], etiolated leaves exposed to light [140], ferritin synthesis in rat liver [146] and fertilization in sea urchin eggs [160]. Specific effects of plant hormones on this process have also been documented. These examples include GA3 effect in barley aleurone cells and spores of Anemia phyllitidis [93,149], IAA effect in pea epicotyls [79] and cytokinin effect in Glycine max [102,103]. In all the above mentioned cases, when external stimuli are provided, the dormant m RNPs get activated and the rate of protein synthesis is enhanced, even in the presence of actinomycin-D, or 5-FU, thereby indicating that the enhanced protein synthesis is independent of new RNA synthesis. It is not clear, whether these external stimuli or added phytohormones act directly on m RNPs or function through other mediating-factors, which may either activate ribosomal machinery or bring about the conformational changes in the secondary structure of m RNPs.
TRANSFER RNA:
Transfer RNA (tRNA) is also synthesized as a large precursor and then processed into smaller products [131]. Transport, splicing and secondary modifications such as methylation, thiolation of pre-tRNAs are some of the important controlling processes, which make tRNAs available, for protein synthesis. Addition of CCA at 3’ terminal of tRNAs in some and activation of amino-acyl tRNAs synthetases are other crucial steps that regulate the translation processes in Pisum sativum seedlings, GA3 is known to enhance aminoacyl tRNA synthetases by 5-8 fold [149]. In early 1960s, auxin was suspected of form a complex with tRNA thus regulating tRNA function [150]. However, it is now considered that this may not be the case [151,152].
As the genetic code is degenerate, it is thought, that multiple iso-accepting species of tRNAs may exert a regulatory role in the translation of mRNAs [153] and the relative changes in the population of such iso-accepting species of t RNAs [154] and the relative changes in the population of such iso-accepting species of tRNA during different stages of development, has been recorded in plants [155,156]. The presence of cytokinins-effect may be mediated through tRNAs [106]. The occurrence of the highly active cytokinin ^6 – de (6-3 methyl-2-butenyl amino purine) adjacent to the A^ base of the anticodon triplet has been demonstrated in yeast serine- tRNA I, II and E. coli- tRNA [157]. Such cytokinin containing tRNAs have been identified in various plants and it is felt, that cytokinins may exert their effect on growth through regulating the translational process. However, recent studies [158] show that there is no correlation between the effect of hormone-induced growth and the presence of cytokinin in tRNA species. Neverheless, there are evidences to show that cytokinins can affect the tRNA activities either by regulating the levels of tRNAs or by secondary modification of tRNA by methylation or by changing amino acyl-synthetase activity [108].
CONTROL OF PROTEIN SYNTHESIS BY PHYTOHORMONES
![]() In radish cotyledons, cytokinin-enhanced
protein synthesis does not require mRNA synthesis de novo [161].
Similarly cytokinin enhances protein synthesis in soybean callus, as early as
30 minutes after treatment [102]. Klambt (1976) [99] has demonstrated that N5
( 2- isopentyl) adenine stimulates cell-free protein synthesis slightly in
systems derived from tobacco pith and corn extracts. Such stimulation is
suggested as due to selective binding of cytokines to a specific protein of the
ribosome. Fox et al. (1975) [162] have identified one low and
one high affinity cytokinin binding site on wheat germ ribosomes. The binding
affinity is lost on boiling the 0.5M KCL wash or on treatment with trypsin,
suggesting that this high affinity receptor is a ribosomal protein. Such cytokinin-binding
protein from tobacco callus is associated with 4OS ribosomes [163]. In animal
systems [164] increased or reduced phosphorylation of ribosomal protein
controls the rate of protein synthesis. Cytokinin is also thought to enhance
protein synthesis through dephosphorylation of ribosomal proteins. Tapfer et
al. (1975) [165] have found that, when cytokinin requiring soybean
callus is transferred to a cytokinin deficient medium, the decreased rate of
protein synthesis is accompanied by increased phosphorylation of some ribosomal
proteins.
In radish cotyledons, cytokinin-enhanced
protein synthesis does not require mRNA synthesis de novo [161].
Similarly cytokinin enhances protein synthesis in soybean callus, as early as
30 minutes after treatment [102]. Klambt (1976) [99] has demonstrated that N5
( 2- isopentyl) adenine stimulates cell-free protein synthesis slightly in
systems derived from tobacco pith and corn extracts. Such stimulation is
suggested as due to selective binding of cytokines to a specific protein of the
ribosome. Fox et al. (1975) [162] have identified one low and
one high affinity cytokinin binding site on wheat germ ribosomes. The binding
affinity is lost on boiling the 0.5M KCL wash or on treatment with trypsin,
suggesting that this high affinity receptor is a ribosomal protein. Such cytokinin-binding
protein from tobacco callus is associated with 4OS ribosomes [163]. In animal
systems [164] increased or reduced phosphorylation of ribosomal protein
controls the rate of protein synthesis. Cytokinin is also thought to enhance
protein synthesis through dephosphorylation of ribosomal proteins. Tapfer et
al. (1975) [165] have found that, when cytokinin requiring soybean
callus is transferred to a cytokinin deficient medium, the decreased rate of
protein synthesis is accompanied by increased phosphorylation of some ribosomal
proteins.
In soybean hypocotyls, a parallel situation is found where Auxin causes increased protein synthesis in a cell-free protein synthesizing system [166]. Travis et al. (1976), have proposed, that the polysome formation is dependent on ribosome activation as well as mRNA supply. In the same system they have also observed the incorporation of labeled amino acids into 3 ribosomal proteins, one of which shows several fold higher incorporation that that of the control. However, ribosomal dissociation studies clearly indicate that these auxin induced proteins are associated with 60S ribosomal sub-units. Evidence for hormone-induced protein synthesis, independent of mRNA synthesis, is also found in Pea epicotyls for [99]. Here, auxin has been found to activate the mRNA specific for cellulase at very early periods, when there is no increased mRNA synthesis. The poly (A) – RNA isolated from treated segments, on translation in vitro, shows enhanced synthesis of cellulase. The poly(A) – RNA from control plants hardly codes for any cellulase. The above results suggest that the auxin perhaps activates the preexisting, dormant mRNAs into active translatable form of mRNA.
Thus, the evidences presented above indicate that most of the growth promoting hormones increase protein synthesis through increased mRNA synthesis as well as through activation of dormant mRNAs or and the ribosomal system. Such dual action of phytohormones is well documented (165 and 166) ABA, in general, appears to exert an inhibitory effect on the synthetic processes. Although, ABA inhibits light-promoted unrolling and greening of etiolated leaves in wheat, it does not prevent light induced polysome formation [167]. In etiolated leaves of barley seedlings, ABA not only reduces the amount of polysome formation in the dark, but also inhibits light-induced increase in polysome size [168]. In barley aleurone cells, GA3 – INDUCED a-amylase synthesis is inhibited by ABA and this inhibition is prevented by the addition of cordycepin. This observation indicates that ABA has both transcriptional as well as translational control [169]. In this context, increase in UMP-rich RNA component, in response to ABA treatment, is highly significant. Heywood et al. (116) have isolated a translational control RNA of mol.wt. 10,000 daltons, which is rich in UMP. This translational control RNA is believed to play a significant role in translational control where it inhibits translation of mRNAs, probably by pairing with poly-AMP segments of mRNAs [116 &170]. This observation correlates well with the effects of ABA on growth inhibition in plants.
PHYTOHORMONES AND RNA – PROTEIN TURNOVER
The regulation of RNA and protein turnover is another important facet of cellular differentiation. Phytohormones are known to regulate such events. ABA, when applied to excised barley leaves, enhances chromatin bound nuclease and deoxyribonuclease (DNase) activities [180]. In excised Avena leaves, it affects one out of the four nuclease isoenzymes [171]. Similarly in lentils (Lens culnaris), ABA reduces the levels of RNA and also inhibits growth [172]. However, in maize coleoptiles, ABA suppresses the incorporation of radioactive precursors into RNA without affecting the levels of RNase for 8 hours [173].
Auxin is also implicated in suppressing the RNase activity in number of cases [174,175]. However, no direct relationship between nuclease levels and RNA stabilization has been established. In pea epicotyls, IAA elicits a large increase in membrane-bound RNase, which in turn is suppressed by kinetin [176]. Similarly, a correlation has been established between nucleases and auxin treatment in wheat root callus cultures [177].
In senescing leaf tissues, increased activities of RNase and DNase are correlated well with the decreased content of RNA and yellowing of the leaf. Cytokinins suppress such activities and delay senescence, which is popularly known as Richmond and Land effect [178-180]. In the absence of a “water stress” on leaves RNase activity is suppressed by kinetin but the same hormone under “water stress” condition enhances RNase activity [181].
In mustard and corn leaves, during senescence, kinetin and 6-benzylaminopurine retard the loss of radioactive label from prelabeled proteins [182, 183]. In wheat leaves, cytokinin suppresses proteolysis of RuDP-carboxylase and this is prevented by CHI, suggesting that the proteolytic enzymes are being synthesized de novo during senescence [184].
During differentiation in Dictyostelium (from plasmodia stage to sporangium) [144], blue-green algae (vegetative to heterocyst) [185], and soybean callus cells (non-dividing to dividing state) [103], appearance and disappearance of some specific proteins have been noticed. This selective appearance and disappearance of certain proteins during developmental process may involve differential expression of genes. Such transitory proteins may play a very important role in morphogenesis.
ADVENTITIOUS ROOT INITIATION AS A MORPHOGENETIC SYSTEM
Introduction:
In the present investigation, hormone induced root initiation in the hypocotyls tissue of Phaseolus vulgaris Linn. It is used as a model system, to study the molecular events involved in the differentiation process. A brief review of the literature on adventitious root formation is therefore presented here.
The root that develops from any part of the plant body other than the radicle is called an adventitious root. Duhmel du Monceau (1758) for the first time explained adventitious root formation in stem as due to the downward movement of sap. Sachs (1880) postulated the existence of a highly potent root forming substance originating from leaves [186]. Van Tieghen and Douliot (1888) observed adventitious root formation in 67 dicotyledonous and 21 monocotyledonous families [187]. In 1933, Bouilleune and went propounded a hypothesis that “Rhizocauline”, a specific factor, is responsible for root initiation [188]. Went and Thimann (1937) observed for the first time that auxin, in addition to its growth-promoting effect, also induces root initiation in stem outings. Thus, a practical use was also found for the auxin [189].
Anatomical studies:
Previous anatomical studies reveal that during root initiation, meristematic activity is found in the region of endodermis, pericycle, especially over the vascular bundles [190-192]. However, there are many examples of adventitious roots originating from phloem parenchyma or phloem ray cells. It is also not uncommon to find roots arising from tissues such as primary xylem parenchyma, callus etc. [193]. The common observation of adventitious roots arising near vascular bundles, prompted Priestly and Swingle (1929) to suggest that nutrient supply may be a factor determining the site of root formation (194).
In recent years, pea epicotyls (Pisum sativum) and hypocotyls of Glycine max (soybean) have been used as systems for studying the effect of plant growth substances in the regulation of cambial activity and organization of root primordial in plants. Excised or intact hypocotyls, epicotyl segment and callus in response to auxin treatment, undergo dedifferentiation leading to the formation of root primordial. Preliminary anatomical studies have shown that parenchymatous cells, in the cortex of 24 hr auxin treated soybean hypocotyls and pea epicotyls, appear to be swollen and exhibit few cell divisions [72,195,196]. After 48 hr the number of cell divisions increase and disintegration of many cortical cells sets in, leaving lacunae. Root primordium originates from the cambial cells near vascular tissues and become recognizable after 3 days of hormone treatment. The primordial cells continue to divide and occupy the lacunae. In 4-5 days the new roots break through the epidermal layers of the segments.
Factors other than phytohormones:
Many environmental factors such as water supply, temperature, sunlight, acidity of the soil, etc. are known to affect root initiation. Young leaves promote root initiation but old leaves are found to inhibit them. Roots themselves inhibit new root formation, perhaps as a result of the production of some inhibitors [197].
The role of organic micronutrients is new root formation is significant. Adenine, arginine, aspargine and uracil promote root initiation [197] but guanine inhibits root formation in Phaseolus mungo [198]. Among other organic micronutrients, nicotinamide, niacin, thiamin (vitamin B1), vitamin K and vitamin H and pyridoxyl enhance root formation only in the presence of auxin [197].
It is also interesting to note, that some cortisols stimulate adventitious root formation in the intact hypocotyls segments of mung beans [199]. The cortisol sensitive phase during root initiation is between 2nd and 4th day of treatment. However, cortisols in the presence of auxins, stimulate root initiation, but inhibit growth of the roots [200]. Recently it has been reported that vitamin D and its analogues by themselves promote root formation slightly, but in the presence of IBA, IAA or NAA, root production is greatly enhanced [201].
Genetics of root formation:
Zobel (1975) has described mutants in tomato with respect to root formation. These mutants are referred to as rosette (ro), lateral-les (dgt) and dwarf (drf), of which r has been established as adventitious rootless mutant [202]. The diploid chromosome number of these plants is 24 (2n = 24). Breeding experiments and genetic analysis have revealed that ro gene is located on the second chromosome and follows simple Mendelian inheritance. Hence, it is suggested that a single pair of alleles controls adventitious root formation.
Plant hormone interactions:
The most effective adventitious root forming substances known are IBA and NAA [188]. Staurt (1938) demonstrated that excised stem cuttings of Phaseolus mobilize carbohydrate reserves as soluble sugars within 24 hr of auxin treatment [203]. Altman and Wareing (1975) obtained similar results using [14 C]-CO2 in Phaseolus vulgaris cuttings [204]. However, the application of exogenous sugars could not replace the requirement of auxins for new root formation [205].
Ethylene stimulates root initiation in many leafy stem cuttings [206], but there are also cases where ethylene has no promotive effects [207,208]. Batten et al. (1978) have claimed that there is no correlation between the effectiveness in rooting and induction of ethylene production by a variety of auxins. Application of ethylene has no effect on root initiation and it proceeds normally even in the presence of 7% carbon dioxide or in hypobaric conditions [209]. In this context, it is pertinent to note, that in the hypocotyls of mung bean, IAA induces a specific protein, which in turn inhibits IAA induced ethylene production [210].
While ABA exhibits both stimulatory and inhibitory effects on new root formation [211,212], cytokinins and GA3 in general are inhibitory [213-215]. However, it has been suggested that the early stages of root formation are most sensitive to cytokinin and GA3 inhibition, but under certain conditions GA3 promotes root formation in Citrus embryoids [216]. Verga et al. (1974) have found that pretreatment of leaves with GA3, before excision, enhances adventitious root initiation. This effect is believed to be due to GA3 induced enhancement of auxin synthesis [217].
In callus cultures, in general, root formation is dependent on the ratio between auxin and cytokinin concentration. While higher ratios promote root formation, a lower ratio inhibits this process but induces shoot primordial. However, at intermediate ratios, both roots and shoots are produced. Hence, it is deduced that the relative concentration of auxin to cytokinin is very critical in the differentiation and development of organs such as roots and shoots [4] Fig.2.
BIOCHEMICAL EVENTS:
During adventitious root initiation in the epicotyls of pea and hypocotyls of soybean, increase in DNA, RNA and protein contents has been observed. After 3 days of auxin treatment, cellulase activity increases 30-40 fold, pectinase 7-8 fold, pectin esterase and b 1 -> 3 Gluconase by 3-fold [195]. Similarly, during rhizogenesis, in detached cotyledons of mustard (Sinapis alba L.) increase in RNA and protein contents have been noticed [218]. Some evidence has been presented to show that the increase in cellulose activity is totally abolished, if the segments are treated with actinomycin D or azaguanine or puromycin along with the auxin. Hence, it is discerned that the continued synthesis of RNA and protein is essential for the increase in the activity of cellulase [196].
Trewavas (1976) has documented some interesting observations on the changes in nucleic acid metabolism and protein synthesis, specifically cellulose, during adventitious root initiation in pea epicotyls and soybean hypocotyls [196] (Fig. 3) In epicotyls of pea plant, over a 3 day period, auxin causes an increase in DNA and protein contents by 2.5 fold and RNA content 4.0 fold. In soybean hypocotyls, a 4-5 fold increase in DNA and protein contents and a 10-foldincrease in RNA content has been observed. Under similar conditions, actinomycin D and puromycin inhibit the auxin induced increase in DNA, RNA and protein contents.
In the presence of auxin, the ratio of microsomal RNA to soluble RNA is 6.9 compared to the control value of 4.8. These changes are attributed to auxin induced differential turnover of RNA. Furthermore, labeling studies have indicated that initial lag period for auxin mediated increase in RAN synthesis is l hr in pea segments and 3 Hr in soybean. After several hours of treatment with auxin, increased labeling has been found in rRNA and tRNA [72,151].
Using in vitro transcriptional assay system and DNA-RNA hybridization techniques, O’ Brien et al. (1968) and Verma et al. (1975) have demonstrated qualitative and quantitative changes in RNA [79, 83]. This implies selective unmasking of the genome, which correlates well with the time point at which cell divisions start.
In a time-course study in pea segments, Davies et al. (1963) have detected a further shift in the labeling pattern of the polysomal populations. At the end of the third hour the proportion of polysomes containing 10 ribosomes or more has increased 3 fold. Ribosomal populations double within 6-10hours. After 10 hr of treatment, the ratio of polysomes to monsomes reaches a value of 9, compared to 0.5 in control tissue [219].
In soybean hypocotyls a shift from monsomes to polysomes is noted after 3 hr of auxin treatment and this is sensitive to actinomycin D, but not to 5-FU, 6-methylpurine in polysome size is independent of ribosomal RNA synthesis, but dependent upon the continued synthesis of tightly bound (TB) and DNA-like RNA (D-FNA). It has been further demonstrated that the increase in protein synthesis by individual polysomes in vitro is due to the increased activation of chain initiation, probably through the synthesis of 3 specific 60s ribosomal proteins [77,166]. Figure 2 and Figure 3.
The present investigation has been undertaken to study (1) the early molecular events leading to redifferentiation of pericyclic cells into root primordial; (2) the identification of specific gene products essential for adventitious root initiation; (3) a delineation of sequence of events at molecular level leading to the emergence of root initials. Adventitious root initiation in hypocotyls segments of Phaseolus vulgaris Linn. In response to the hormone IBA treatment has been used as the model system.

Fig.2: Interaction between Auxins and Cytokinins during Organ differentiation in callus cultures. Open arrows indicate the promotion of organs and closed arrows indicate inhibition.

Fig.3: time of initiation of metabolic changes induced by IAA after addition to Soybean hypocotyls or pea epicotyls.
Discussions
Plant hormones control the process of growth and differentiation in a variety of plant cells. The underlying hormone induced changes in gene expr3ession are responsible for the subsequent phenotype effects observed. It can as well be that the hormone triggers off changes primarily at the cytoplasmic level which brings about an ordered sequence of biochemical changes at the genetic level, committing the cells to a particular course of the differentiation process. A delineation of the sequence of biochemical changes involved, is of considerable importance to understand the molecular basis of the complex process of differentiation.
In the present study, Indole butyric acid induced root formation in the hypocotyls segments of the Phaseolus vulgaris L. has been used as a model system. First of all, it has been established that external addition of IBA is absolutely necessary for root formation in hypocotyl segments maintained in Hoagland’s medium. Exposure of the segments to an IBA concentration of 8-10 mg/ml for a period as short as 10 min is sufficient for the production of roots, counted after a period of 72 hours.
At the anatomical level, it has been found that only the pericyclic cells that are locate above the split exarch xylem strands respond to the auxin stimulus. Within 24 hr of auxin treatment these cells show enlargement of the nuclei and nucleoli. By 36-48 hr these cells show directional and polarized cell division and manifest emergence of root primordial in four rows.
At the biochemical level, IBA treatment has been found to lead to increased protein synthesis as early as 30 min after hormone tre3atment. This very early increase in protein synthesis is not dependent on a concomitant increase in RNA synthesis. The observation that there is no change in RNA synthesis at this time point as well as that actinomycin-D fails to inhibit the hormone induced increase in very early protein synthesis, substantiate this point. This effect of the hormone appears to be at the translation level. The cytosolic fraction (S 150) isolated from hypocotyls treated with IBA for 30 min stimulates more protein synthesis than that of the control cytosol in cell-free protein synthesis experiments. At the same time, ribosomes isolated from such hormone-treated hypocotyls are also more active than control ribosomes. The higher activity of the ribosomes from the hormone treated hypocotyls can at least be in part due to activation of the messenger RNA. This is proven by the fact that at the same concentration, poly (A)-containing RNA fraction isolated from hormone-treated hypocotyls is more potent than the poly(A)-containing RNA fraction from control segments, in stimulating protein synthesis in the wheat germ cell-free system. Thus, the hormone-induced RNA-independent increase in very early protein synthesis appears to be due to an activation of cytoplasmic / ribosomal protein factors as well as a probable change in the secondary structure of the messenger RNA.
The subsequent increase in hormone-induced protein synthesis, detectable at 2 hr after IBA-treatment, is mediated by a concomitant increase in RNA synthesis. This event, termed as hormone-mediate early protein synthesis, is susceptible to actinomycin-D treatment. An increase in total as well as poly (A)-containing RNA synthesis can be demonstrated at this time. Quantification of the messenger content in the wheat germ cell-free system reveals that the messenger content increases by 2-4 fold between 6 hr to 48 hr after IBA –treatment of the hypocotyls.
The involvement of IBA-induced very early protein synthesis in root formation has been investigated with the use of cycloheximide to block the very early protein synthesis. This leads to a striking delay in root initiation and a substantial decrease in the number of roots initiated. In these experiments, cycloheximide has been added only for a brief period to inhibit the IBA-induced very early protein synthesis and the rate of total protein synthesis recovers back to normal levels in about 2 hours. Surprisingly however, cycloheximide and IBA-pretreated hypocotyls also show a decreased rate of RNA synthesis and the recovery of RNA synthesis is itself a slow process, having a lag period of 12 hours. Thus it appears that the activation of very early protein synthesis by IBA may lead to activation of RNA synthesis which may in turn code for some of the hormone induced early proteins.
An analysis of the early proteins coded for by the hypocotyls messenger RNA in the wheat germ system reveals that IBA induces both quantitative and qualitative changes. A significant increase in high molecular weight proteins (70,000-1, 75,000), a species with 55,000 molecular weight and low molecular weight proteins (20,000 – 5,000) have been observed under conditions of IBA treatment. The 55,000 molecular weight species, whose level increases strikingly in the membrane fraction of IBA-treated hypocotyls, has been identified as tubulin based on electrophoretic mobility, involvement in the polymerization process and immunological criteria. IBA enhances tubulin synthesis. Another interesting feature is that IBA is capable of activating tubulin polymerization IN VITRO. This effect may underlie the immediate hormone effect in cell elongation and may also have a role is the subsequent differentiation process. The involvement of tubulin and its polymerization in IBA-induced root initiation is proved by the inhibitory effects of colchicines and cytochalasin-B on this process.
The concept of a target cell for a hormone is very well accepted in animal cells. However, in plant cells a few hormones are able to bring about diverse effects in a variety of cells depending on the concentrations of the individual hormone species. Thus, the process of differentiation in a plant cell is esse3ntially governed by hormone-hormone interaction. In order to understand this process, the effect of hormone-hormone interaction in adventitious root initiation has been examined. It has been -found that 6 – benzylaminopurine at concentrations above 1 mg/ml, prevents IBA-induced root initiation in the hypocotyls tissue of P. vulgaris. Thus, a study of this interaction at the biochemical level can provide an insight into the key steps involved in the differentiation process. 6-Benzylamino purine has been found to elicit effects on protein and RNA synthesis grossly similar to those of IBA, expect that the BAP-induced RNA synthesis is manifested with a lag of 12 hours. However, the IBA + BAP effects are not additive. Again, IBA + BAP treatment enhances cell division strikingly but there is no cell polarization and formation of root primordials. Tubulin synthesis takes place in BAP as well as IBA + BAP treated hypocotyls. But in BAP or IBA + BAP treatments there is a decrease in messenger RNA coding for the high molecular weight early proteins detected in IBA-treatment. Thus, it appears possible that these high molecular weight proteins may influence tubulin polymerization into specific microtubular assemblies governing the differentiation process. It is known that microtubule assembly involving tubulin polymerization is governed by high molecular weight proteins such as MAPs and TAU. Further studies are needed to examine the mode of IBA-BAP interaction. Based on the present study the following sequence of biochemical events leading to root initiation by IBA in the hypocotyls has been proposed (Fig. 33A and B)


Acknowledgements and References
It is with deep sense of gratitude I record my appreciation to Prof. G. Padmanabhan for his guidance, help and encouragement during the course of this investigation. Especially, I wish to acknowledge the latitude I enjoyed under his tutelage in planning and executing the experimental work. I express my gratefulness to Prof. S. Mahadevan for the guidance and suggestions offered from time to time.
I am indebted to Dr. Lakshmanan for his expert help in conducting some experiments concerned with Tubulin. I gratefully acknowledge the ready and voluntary help rendered in my experimental work by Dr. M.R.S. Rao, Dr. K.S. Bhat, Mr. Ashwini Kumar, Mr. Ravi Shankar and Mr. S. Parimoo. With a sense of appreciation I thank Miss. Dr B.H.M Nijalingappa, Usha, Dr. M. Seenappa, Mr. Manu Kumar, Mr. Shashidhar, Mr. Jagadish, and Prabhu for their help in preparing the manuscript. I also thank many of my friends and colleagues for helping me in the course of this investigation in one-way or the other.
During the course of my research, the expert assistance by Mr. Shivakumar has been gratefully acknowledged. I also thank Mr. Gopinath, Mr. Gururaj, Mr. Nagendranath and Mr. Padki. Mr. Rama has been a great help to me for which I am highly obliged.
I thank Dr. Kamath for good Typographic work and Mr. Narayan for his neat drawings. My thanks are also due to Mr. B.R. Seshadri for his alertness and excellent typing of the thesis.
I am highly indebted to my institution – The National College, Basavanagudi, Bangalore, which has given me a foothold and a standing. I gratefully acknowledge Dr. H.N’s encouragement and constant remainding me of the importance of higher academic achievement. I thank Prof. M.S. Venkata Rao, the Principal of my College, for giving me an opportunity to work for Ph.D. With deep sense of gratitude I acknowledge the financial help by The University Grant Commission, the Director of Collegiate Education, Govt of Karnataka.
I have many warm memories of my association with this Department of Biochemistry and its teaching staff. It is an unforgettable experience to be a student in this department, which I proudly cherish in my heart.
I am highly grateful to my wife Smt. Jyothi and to my children Sanju and Rahul for their forbearance during my three years of Ph.D. work. They have truly shared my agony and ecstacy with good spirit.
I still remember the remarks made by a Chinese Scholar of 13th Century – “Were I to await perfection my book would never be finished.” I am in the same predicament.
G.R. KANTHARAJ
REFERENCES:
1. Darwin, C. and Darwin, F. (1880) the power of movements in plants, John Murray, London.
2. Kogl, F., Haagen Smit, A.J. and Erxleben, H. (1934) Z. Physiol. Chem. 214, 241.
3. Aldus, L.J. (1972) Plant growth Substances, Vol. I, Leonard Hill, London, PP.39-40.
4. Miller, C.O. (1961) Ann. Rev. Plant Physiol. 12, 395.
5. Milborrow, B.V. (1974) Ann. Rev. Plant Physiol. 25,259.
6. Lance, B., Reid, D.M., Thorpe, T.A. (1976) Physiol. Plant. 36, 287
7. Engelke, A.L.,Hamzi, H.Q. and Skoog, F. (1973) Amer. J. Bot. 60,491.
8. Letham, D.S., HIGGINS, T.J.V., Goodwin, P.b. and Jacobsen J.V. (1978) in: Phytohormones and related compounds, Vol. I, Elsevier, North Holland, pp.11-13.
9. Quatrano, R.S. (1978) Ann. Rev. Plant Physiol. 29 487.
10. Hopler, P.H. and Palevitz, B.A. (1974) Ann. Rev. Plant. Physiol. 25, 309.
11. Edelman, G.M. (1976) Science 192, 218.
12. Nicholson, G.L. (1976) Biochim. Biophys. Acta 457, 57.
13. Naccitelli, R. (1978) Dev. DBiol. 62, 13.
14. Peng, H.B. and Jaffe, L.F. (1976) Planta 133, 57.
15. Laderbetter, M.G. and Porter, K.R. (1963) J. Cell. Biol, 19, 239.
16. Witman, G., Carlson, K., Berliner, J. and Rosenbaum, J.L. (1972) J. Cell. Biol. 54, 507.
17. Dentles, W.L., Grannezz. S. and Rosenbaum J.L. (1975) J. Cell. Biol, 65, 237.
18. Weingarten, M.W., Lockwood, A.H., Hwo, S.Y. and Kirschner, M.W. (1975) Proc. Nat. Acad. Sci. U.S.A. 72 , 1858.
19. Sloboda, R.D., Rudolph, S.A., Rosenbaum, J.L. and Greengard (1975) Proc. Nat. Acad. Sci. USA. 72, 177.
20. Behnke, 0. (1971) Int. Rev. Expt. Pathol. 9, 1.
21. Pickett-Heaps, J.D.(1969) Cytobios. 1, 257.
22. Marchant, H.(1978) Expt.cell res. 115 ,25.
23. Lloyd, C.W., Slbas, A.R., Powel, A.J., Macdonald, D. and Badley, R.A. (1979) Nature 279, 239.
24. Shbaok, H. (1974) Plant. Cell Physiol. 15, 255.
25. Sawhney, V.K. and Srinivastava, L.M. (1974) Can. J. Bot. 52, 259.
26. Lawson, V.R. and Weintraub, R.L. (1975) Plant Physiol. 55, 1062.
27. Kieth, B. and Srivastava, L.M. (1978) Planta 139, 301.
28. Helper, P.K. and Pelvitz, B.A. (1974) Ann. Rev. Plant. Physiol. 25, 309.
29. Pope, D.G., Thorpe, J.R., Al-Azzawi and Hall, J.L. (1979) 144, 373.
30. Chrispels, M.J. (1976) ann. Rev. Plant Physiol. 27, 19.
31. Atsmon, D. (1972) in: Plant growth substances, D.G. Carr (ed), Springer/Verlag, Berlin, pp.222-227.
32. Jacobsen, J.V. and Higins, T.J.V. (1978) in: Phytohormones and related compounds, Vol. I, Letham, D.S., Goodwin, P.B. and Higgins, T.J.V. (eds), Elsevier-North-Holland, pp.515-528.
33. Wardell, W.H. (1975) Plant Physiol, 56, 171.
33a. Libbenga, K.R. and Torry, T.J., (1973) Amer. J. Bot. 60, 293.
34. Uffler, H.R., O’Brien, T.J., Glover, D.V. and Cherry, J.H. (1971) Plant Physiol. 48, 43.
35. Degani, Y. and Atsmon, D. (1970) Nature 228. 554.
36. Degani, Y. and Atsmon, D. (1970) Expt. Cell Res. 61, 226.
37. Holm, J.H., O’Brien, T.J., Key, J.L. and Cherry, J.H. (1970) Plant Physiol. 45, 41.
38. Apelbaum, A. and Barg, S.P. (1972) Plant Physiol. 50, 117.
39. sarrouy-Balat, H., Delseny, M. and Julien, R. (1973) Plant Sci. Lett. L, 287.
40. Simond, A. (1971) Can. J. Bot. 49, 1541.
41. Jouanneau, J.P. and Tandeau de Marsac N. (1973) Expt. Cell Res. 77, 167.
42. Jouanneau, J.P. (1975) Expt. Cell Res. 91, 184.
43. Kiper, M., Bortels, D., Hozfeld, F. and Richter, G. (1979) Nucl. Acid Res. 6, 1961.
44. Goldberg, R.B., Hoschek, G. and Kamalay, J.C. (1978) Cell 14, 123.
45. Britten, R.J. and Davidson, E.H. (1969) Science 165, 345.
46. Kavoor, a. and Melet, d. (1972) Symp. Biol. Hung. 13, 29.
47. Kessler, B. (1973) in: Biochemistry of gene expression in higher organisms, J.K. Pollack and J.W. Lee (eds), Australia and New Zealand Book Co., Sydney, pp.333-356.
48. Nagl, W.and Rucker, W. (1976) Nuc. Acid Res. 3, 2033.
49. Nagl, W. (1975) Prog. Bot. 37, 186.
50. Cleland, R.E. (1975) Planta 127, 233.
51. Ray, .M. and Dohrmann, U. (1977) Plant, Physical. 59, 357.
52. Kasomo, K. and Yamaki, t. (1976) Plant Cell. Physiol. 17, 149.
53. Cleland, R.E. (1973) Proc. Nat. Acad. Sci. U.S.A. 70, 3092.
54. Wardrop, A.J. and Polya, G.M. (1977) Plant Sci. Lett. 8, 153.
55. Ihl, M. (1976) Planta 131, 223.
56. Hardin, J.W., O’Brien, T.J. and Cherry, J.H. (1970) Biochim. Biophys. Acta 224, 667.
57. Hardin, J.W., Cherry, J.H,, Morre, D.J. and Lembi, C.A. (1972) Proc. Nat. Acad. Sci. U.S.A. 69, 3146.
58. Lembi, C.A. (1972) Proc. Nat. Acad. Sci. U.S.A. 69, 3146.
59. Mathyse, A.G. and Phillips, C. (1969) Proc. Nat. Acad. Sci. U.S.A. 63, 897.
60. Mondal, H., Mandal, R.K. and Biswas, B.B. (1972) Nature 240, 111.
61. Biswas, B.B., Ganguly, A., Das., and Roy, P. (1975) in: Regulation of growth, differentiation and function in eukaryotic cells, Talwar, G.P. (ed), Raven Press, New York, pp.461-477.
62. Fellenberg, G. (1971) Planta 100, 347.
63. Bamberger, E.S. 1971) Phytochem. 10, 957.
64. Grieshaber Scheubel, D. and Fellenberg, G. (1972) Z. Pflanzenphysiol. 66, 106.
65. Cohen, M.E. and Klein Smith, L.J. (1976) Biochim. Biophys. Acta (Amst.) 435, 159.
66. Klein Smith, L.J., Stein, J. and Stein, G. (1976) Proc. Nat. Acad. Sci. U.S.A. 75, 1174.
67. Spelsberg, T.C., Webster, R.P. and Picker, G.M. (1976) Nature (Lond.) 261, 65.
68. Spelsberg, T.C. and Sarkissian, I.V. (1968) Phytochemistry 7, 2083.
69. Murray, M.G. and Key, J.L. (1978) Plant Physiol. 61, 190.
70. Tissere, M., Penon, P., Van Huystee, R.d., Azon, Y. and Richard, J. (1975) biochim, Biophys. Acta (Amsterdam) 402, 391.
71. Silberger, J. and Skoog, F. (1953) Science 118, 443.
72. Trewavas, A.J. (1968) Arch. Biophys. And Biochim. 123, 324.
73. Key, J.L. and Ingle, J. 1964) Proc. Nat. Acad. Sci. U.S.A. 52, 1382.
74. Nooden, L.D. (1968) Plant Physiol. 43, 140
75. Trewavas, A.J. (1968) Phytochemistry 7, 673.
76. Fites, R.C., Hanson, H.B. & . Slife, F.W. (1969) Bot. Gaz. 130
77. Travis, R.L., Anderson, J.M. and Key, J.L. (1973) Plant Physiol. 52, 608.
78. Davies, E. (1976) Plan, Physiol. 57, 516.
79. Verma, D.P.S., Maclachlan, G.A., Byrne, H. and Ewings, D. (1975) J. Biol. Chem. 250, 1019.
80. Teissere, M., Penon, P., Ricard, J. and Rattle, G. (1972) FEBS Lett. 23, 65.
81. Kiper, M., Bortels, D., Herzfeld, F. and Richter, G. (1979) Nucleic Acid Res. 6, 1961.
82. O’Brien, T.J., Jarvis, B.C., Cherry, J.H. and Hanson, J.B. (1968) in: Biochemistry and Physiology of Growth Substances, F. Wieghtman and G. Setterfield (eds), Runge Press, Ottawa, pp.747-759.
83. O’Brien, T.J., Jarvis, B.C., Cherry, J.H. and Hanson, J.B. (1968) Biophys. Acta (Amst.) 169, 35.
84. Konjevic, R., Grubisic, Markovic. R. and Petrovic, J. (1976) Planta 131, 125.
85. Nadeau, R. and Rapport, L. (1974) Proc. Nat. cad, Sci. U.S.A. 71, 3253.
86. Johri, M.M. and Varner, J.E. (1968) Proc. Nat. Acad. Sci. U.S.A. 59, 269.
87. Kende, H. and Gardner, G. (1976) Ann. Rev. Plant Physiol. 27, 267.
88. Kessler, B. (1971) Biochim, Biophys. Cta (Amst.) 232, 611.
89. Snir, I. And Kessler, B. (1975) Physiol. Plant. 35, 191.
90. Jacobsen, J.V. (1977) Ann. Rev. Plant Physiol. 28, 537.
91. Varner, J.E. and Chandra, G. (1964) Proc. Nat. Acad, Sci. U.S.A. 52, 100.
92. Ho, D.T.H. and Varner, J.H. (1974) Proc. Nat. Acad. Sci. U.S.A. 71, 4783.
93. Higgins, T.J.V., Zwar, J.A. and Jaccbsen, J.V. (1976) Nature 260, 166.
94. Carlson, P.S. (1972) Nature 237, 39.
95. Rodaway, S. and Kende, H. (1978) Plant Physiol. 61, Suppl. 188, p.34.
96. Wasilewsky, L.D. and Kleczicowski, K. (1976) Eur. J. Biochem. 66, 405.
97. Mathyse, A.G. and Philips, C. (1969) Proc. Nat. Acad. Sci. U.S.A. 63, 897.
98. Klambt, D. (1974) Planta 118, 7.
99. Klambt, D. (1976) Plant Cell Physiol. 17, 73.
100. Shannon, J. and Key, J.L. (1963) Plant Physiol. 38, Suppl. XIV.
101. Vanderhoef, L.N. and Key, J.L. (1988) Plant Cell Physiol. 9, 343.
102. Maab, H., and Klambt, D. (1977) Planta 133, 117.
103. Tepfer, D.A. and fosket, D.E. (1978) Dev. Biol. 62, 486.
104. Bick, M.D.H., Liebke, H., Cherry, J.H. and Strehler, B.Z. (1970) Biochim. Biophys. Acta (amst.) 204, 175.
105. Venkataraman, R., Delec, P., Anderson, M.B. and Cherry, J.R. (1970) Plant Physiol. Suppl. 45, 9.
106. Kende, H. (1971) Int. Rev. Cytol. 31, 301.
107. Skoog, F. (1973) Biochem. Soc. Symp. 38, 195.
108. Burrows, W.J. (1975) Curr. Advance Plant. Sci. No.21, 837.
109. Horton, R.F. and Osborne, D.J. (1969) Nature 214, 1086.
110. Morre, D.J. (1968) Plant Physiol. 43, 1545.
111. Hulme, A.C., Rhodes, M.J.C. and Woolforton, L.S.C. (1971) Phytochemistry 10, 749.
112. Pearson, J.A. and Warieng, P.F. (1969) Nature 221, 672.
113. Benson, R.H. (1971) Plant Physiol. 47, 5.
114. Stain, G.C., Mullinx, K.R. and Bogorad, L. (1971) Proc. Nat. Acad. Sci. U.S.A. 68, 2647.
115. Villiers, T.A. (1968) Planta 82, 342.
115a. Khan. A.A. and Anojulu C.C (1970), B.B.R.C, 38, 1069
116. Heywood, S.M. and Kennedy, D.S. (1976) Biochemistry 15, 3314.
117. Dobrzanska, M. and Bucjpwocz, J. (1976) Biochim. Biophys. Acta (Amst.) 432, 73.
118. Zwar, J.A. and Jacobsen, J.V. (1972) Plant Physiol. 49, 1000.
119. chrispeels, M.J. and Varner, J.E. (1966) Nature 212, 1066.
120. Buller, D.C., Parker, W. and Grant Reid, J.S. (1976) Nature 260, 169.
121. Perry, R.P. (1976) Ann. Rev. Biochm. 45, 605.
122. Monneson, A. and Beranrd, W. (1969) J. Cell Biol. 80, 266.
123. Parvinen, M. AND Parvinea, L.M. (1979) J. Cell Biol. 80, 621.
124. Grierson, D. and Loening, U.E. (1974) Eur. J. Biochem. 44, 501.
125. Grierson, D. and Covey, S. (1976) Planta 130, 317.
126. Brawermen, G. (1974) Ann. Rev. Biochem. 43, 621-642.
127. Schmid, B.D., Siegel, N.R. and Vanderhoef, L.N. (1975) Plant PHYSIOL. 55, 277.
128. Highfield, F.E. and Ellis, R.J. (1978) Nature 271, 420.
129. Ro-Choi, T.S., Reddy, R., Cho V.C., Raj, N.B. and Henning (1974) Fed. Proc. 33, 1548.
130. Muthukrishnan, S., Both, G.W., Furuichi, Y. and jShatkin, A.S. (1975) Nature 255, 33.
131. Perry, R.P. 1976 Ann. Rev. Biochem. 45, 605.
132. Furuichin, Y., Morgan, M., Muthukrishnan, S. and Shatkin, A.J. (1975) Proc. Nat. Acad. Sci. USA 75, 362.
133. Lodish, H.F. (1976) Ann. Rev. Biochem. 45, 39
134. Patterson, B.M. and Rosemberg, M. (1979) Nature 279, 692.
134a. Sonenberg, N., Rupprecht, K.M. Hecht, S.M. and Shatkin, A.J. (1979) Proc. Nat. Acad. Scin, U.S.A., 76, 4345,
135. hall, T.C., Ma, Y., Budibinder, B.U., Pyne, J.W., Sun, S.M. and Bliss, F.A. (1978) Proc. Nat, Acad. Scin, U.S.A. 75, 3196.
136. Kozak, M. (1978) Cell 15 , 1109.
137. Aztkhozhin, M.A., Pollimbetova, N.S. and Akhanov, A.U., FEBS Lett., 54,212.
138. Brooker, J.D., Tomaszewski, M. and Marcus, A. (1978) Plant Physiol. 61, 145.
139. Hammerling, (1963) Ann. Rev. Plant Physiol. 14, 65.
140. Giles, A.B., Grierson, D. and Smith. H. (1977) Planta 136, 31.
141. Seibert, G., Loris, K., Zollner, J., Frenzel, B. and Zahn, R.K. (1976) Nucl. Acid. Res. 3, 1977.
142. Raghavan, V. (1977) J. Cell. Sci. 23, 85.
143. Dhindsa, R.S. and Bewly, J.D. (1978) Proc. Nat. Acad Sci. U.S.A. 75, 842.
144. Alton, T.H. and Lodish, H.F. (1977) Cell 12, 301.
145. Bendish, J.W. and Raff, R.A. (1979) Dev. Biol, 67, 99.
146. Zahringer, J., Baliga, B.S. and Munro, H.N. (1976) Proc. Nat. Acad. Sci. U.S.A. 73, 857.
147. Bester, A.J., Kennedy, D.S. and Heywood, S.M. (1975) Proc. Nat. Acad. Scin. U.S.A. 72, 1523.
148. Henshall, J.D. and Goodwin, T.W. (1976) Planta 130, 269.
149. Henshall, J.D. and Goodwin, TOW. (1964) Phytochemistry 3, 677.
150. Armstrong, D.J. (1966) Proc. Nat. Acad. Sci. USA. 56, 64.
151. Key, J.L. and Ingle, J. (1968) IN: Biochemistry and Physiology of Plant Growth Substances, F. Wightman and G. Setterfield (eds), Runge Press, Ottawa, PP.711-722,
152. Davies, P.J. and Galston, A.W. (1971) Plant. Physiol. 47, 435.
153. Gerlinger, P., Lemeur, M.A. and Ebel, J.P (1975) FEBS Lett. 49, 376.
154. Venderhoef, L.W. and Key, J.L. (1970) Plant Physiol 46, 294.
155. Venkataraman, R. and Deleo, P. (1972) Phytochemistry 11, 923.
156. Patel, H.V. and Pillay, D.T.N. (1976) Phytochemistry 15, 401.
157. Skoog, F. and Armstrong, D.J. (1970) Ann. Rev. Plant Physiol. 21, 359.
158. Burrows, W.J. (1976) Planta 130, 313.
159. Bhatt, S.P. and Padayatti, J.D. (1975) Nature 256 , 227.
160. Hille, M.D. and Albers, A.A. (1979) Nature 278, 469.
161. Gordon, M.E. and Letham, D.S. (1975) Aust. J. Plant Physiol. 2, 129.
162. Fox, J.E. and Erion, J.C. (1975) Biochem, Biophys. Res. Commun. 64, 694.
163. Takagami, T. and Yoshida, K. (1977) Platn and Cell Physiol. 18, 337.
164. Ralph, R.K., Mccombs, P.J.A., Tener, G. and Wojcik, S.J. (1972) Biochem. J. 130, 901.
165. Tepfer, D.A. and Fosket, D.E. (1975) Phytochemistry 14, 1161.
166. Travis, R.L. and Key, J.L. (1976) Plant Physiol. 57, 936.
167. Pearson, J.A. and Wareing, P.F. (1970) Planta 93, 309
168. Person, J.A. and Beevers, L. (1970) Plant Physiol. 46, 782.
169. Ho, D.T.H. and Verner, J.E. (1976) Plant Physiol. 57, 175.
170. Heywood, S.M., Kennedy D.S., and bester (1975) Europ.J Biochem, 58, 587.
171. Wbrmingham, B.C. and Maclachlan, G.A. (1972) Plant Phgen, N.V., Udavardy, J., Erdei, S. and /farkas, G.L. (1972) Symp. Biol. Hung. 13, 293.
172. Pilet, P.E. (1970) J. Expt. Bot. 21., 446.
173. Bex, J.H.M. (1972) Planta 103, 1.
174. Truelson, T.A. (1972) Physiol. Plant u20,1112.
175. Pilet, P.E. and Brount, R. (1970) Physiol. Plant. 23, 245.
176. Birmingham, B.C. and Maclachlan, G.A. (1972) Plant Physiol. 49, 371.
177. Udvardy, J., Sivok, B. and Nemet, G. (1976) Z.Pflanzenphysiol. 78. 33.
178. Richmond, A.E. and Lang, A. (1957) Nature 252, 257.
179. Srivastava, B.I.S. (1968) Biochem. Biophys. Res. Commun. 32, 533.
180. Srivastava, B.I.S. (1968) Biochim. Biophys. Acta (amst.) 169, 534.
181. Arad, S., Mizrahi, Y. and Richmond, A.E. (1973) Plant Physiol. 52, 510.
182. Kuraishi, S. (1968) Physiol. Plant. 21, 78.
183. Tavares, J. and Kende, H. (1970) Phytochemistry 9, 1763.
184. Peterson, L.W. and Huffakar, R.C. (1975) Plant Physiol. 55, 1009.
185. Haselkorn, R. (1978) Ann. Rev. Plant Physiol. 29, 319.
186. 186. Giround, R.M. (1969) Can. J. Bot. 47, 687.
187. Van tieghem, M. and Douliot, H. (1888) Ann. Sci. Nat. Bot. Ser. 7-8, 1.
188. J. Van Overbeck (1961) in: Encyclopedia of Plant Physiology, Vol. XIV, W. Ruthland (ed), Springerverlag, Berlin, pp.1145-1146.
189. Went, F.W. and Thiman, K.V. (1937) Phytohormones, Macmillan Co., New York.
190. Christine J. Gorter (1961) in: Encyclopedia of Plant Physiol. Vol. XIV, W. Ruthland (ed), Springer-Verlag, Berlin, pp.1084-1089.
191. Fahn, a. (1967) Plant anatomy, Pergamon Press, Oxford. Pp.256-257.
192. Esau, K. (1953) in: Plant Anatomy, Johwn Wiley (1972 ed.), New York, pp. 481-538.
193. Byrne, J.M., Collins, K.a., Cashau, P.f. and Aung, L.H. (1975) Amer. J. Bot. 62, 731.
194. Priestley, J.H. and Swingle, c.f. (1929) U.S. Dept. Agr. Tech. Bull. 151.
195. Datko, A.K. and Maclachlan, G.A. (1968) Plant Physiol. 43, 735
196. Trewavas, A.J. (1976) in: Molecular aspects of gene expression in plants, Bryant J.A. (ed), academic Press, New York, London, pp.249-298.
197. Dore, J. (1965) in: Encyclopedia of Plant Physiol. XV/2, W. ruthland (ed), Springer-Verlag, pp.48-91.
198. Sheila, B., Bhattacharya, N.C. and Nanda, K.K. (1978) Physiol. Plant 42,
199. Genus, J.M.C. (1978) Phytochemistry 17, 1.
200. Loeys Mieke, E.J. and Genus, J.M.C. (1978) Z. Pflanzenphysiol. 87, 211.
201. Buchala, A.J. and Schmid, E. (1979) Nature 280, 230.
202. Zobel, R.W. (1975) in: The development and function of roots, Torrey, J.G. and Clarkson, D.G. (eds), Academic Press, London, pp.261-275.
203. Stuart, N.W. (1938) Bot. Gaz. 100, 298.
204. Altman, a. and Warieng, P.F. (1975) Physiol. Plant 33, 32.
205. Nanda, K.K. and Jain, M.K. (1971), New Phytol. 70, 945.
206. Cummins, J.N. and Fiorino, P. (1969) Hort. Sci. 4, 339.
207. Shanks, J.B. (1969) Hort. Sci. 4, 56.
208. Mullins, M.G. (1972) in: Plant growth substances 1970, D.J. Carr (ed), Springer-Verlag, Berlin, pp.526-533
209. Batten, D.J. and Goodwin, P.B. (1978) in: Phytohormones and related hormones: A comprehensive treatise, Vol.II, D.S. Letham, P.B. Goodwin and T.J.V. Higgins (eds), Elsevier, North-Holland, pp.137-145.
210. Todaka, I., Watanabe, a. and Imaseki, H (1978) Plant and Cell Physiol. 19, 561.
211. Basu, R.N., Roy, B.N. and Bose, T.K. (1970) Plant Cell Physiol. 11, 681.
212. Pierik, R.L.M. and Steegmans, HH.M. (1975) Hort. Sci. 3,1.
213. Smith, D.R. and Thrope, T.A. (1975) J. Expt. Bot. 26, 193.
214. Eriksen, E.N. (1974) Physiol. Plant 30, 163.
215. Kefford, N.P. (1973) Plant Physiol. 51, 214.
216. Koehba, J., Button, J., Spiegalroy, P. and Bornmann (1974) ann. Bot. (Lond.) 38, 759.
217. Verga, M, and Humphries, E.C. (1974) Ann. Bot. (Lond.) 38, 803.
218. Moore, K.G. and Lovell, P.H. (1972) Physiol. Veg. 10, 223.
219. Davies, E. and Larkins, B.A. (1973) Plant Physiol. 52, 339.
220. Arnold, D. and Joseph, A. (1968) in: Experimental Physiol, Holt-Rinehart and Winston, London, p.265.
221. Lowry. O.H., Rosenbrough, N.J., Farr, A.L. and Randall, R.J. (1957) J. Biol. Chem. 193, 265.
222. Rosen, H. (1957) Arch. Biochem. Biophys. 67, 10.
223. Anderson, J.M., and Key,. J.L. (1971) Plant Physiol. 48, 801.
224. Penman, s. (1966) J. Mol. Biol. 17, 117.
225. Padmanabhan, G., Hendler, F., Patzer, J., Ryan, R. and Rabinowitz, M. (1975) Proc. Nat. Acad, Sci. U.S.A. 72, 4293.
226. Sardana, M.K., Satyanarayana Rao, M.R. and Padmanabhan, G. (1975) Biochem. J. 147, 185.
227. Laemmli, U.K. (1970) Nature 227, 680.
228. Went, F.W. (1934) Proc. K. Ned. Akad. Wet. 37, 1.
229. Galston, A.W. (1948) Amer. J. Bot. 35, 281.
230. Jensen, W.A. (1962) Botanical Histochemistry, Principles and Practice, W.H. freeman Co., Sanfrancisco, pp.55-83.
231. Zwar, J.A., Higgins, T.J.V. and Jacobsen, J.V. (1979) in: The Tenth International Conference on Plant Growth Substances (Abstracts), Madison, Wisconsin, No.109, p.10.
232. Muthukrishnan, S., Maxwell, E.sS. and Chandra, G.R. (1979) International Conference on Plant Growth Substances (Abstracts), Madison-Wisconsin, No.156, p.41.
233. Chakrabarthy, P.K. and Schneider, W.C. (1978) Cancer Res. 38, 2043.
234. Roberts, B.E. and Patterson, B,M, (1973) Proc. Nat. Acad, Sci. U.S.A. 70, 2330.
235. T.J.V. Higgins and J.V. Jacobsen (1978) in: Phytohormones and Related Compounds: A Comprehensive Treatise, Vol. I, D.S. Letham, P.B. Goodwin and T.J.V. Higgins (eds), Elsevier/North-Holland, pp.419-465.
236. Oppenoorth, J.M. (1978) Physiol. Planta 42, 375.
237. Stearns, M.E. and Brawn, D.C. (1979) Proc. Nat. Acad. Sci. U.S.A. 76, 5745.
238. Lai, E.X., Walsh, C., Wardel, D. and Fulton, C. (1979) Cell 17, 867.
239. Cohen, C. (1979) Trends Biochem. Sci. 4, 97.
240. Cohen, C. (1979) Trends Biochem. Sci. 4, 73.
241. Campell, H.H., Garvey, J.S., Cramer, N.E. and Sussdrof, D.H. (1970) in: Methods in Immunology, W.A. Benjamin Inc., New York, p.189.
242. Thomas, P.E., Lu, A.Y.H., Ryan, D., West, S.B., Kawalek, J. and Levin, W. (1976) J. Biol. Chem. 251, 1384.
243. Snyder, J.A. and McIntosh, J.R. (1976) Ann. Rev. Biochem. 45, 699.
244. Weingarten, M.D., Lockwood, A.H., Hwo, S. and Kirschner, M.M. (1975) Proc. Nat. Acad. Sci. U.S.A. 72, 1858.
245. Schroeder, T.E. (1970) J. Embryol. Exp. Morphol. 23, 427.
246. Bonner, W.M. and Laskey, R.A. (1974) Eur. J. Biochem 46, 83.
247. Callissano, P., Monaco, G. Levi, A., Menesinichen, g.M., Chem, J.S. and Levi-Montalcini, R. (1976) in: Contractile systems in non-muscle tissues, Perry, S.V. el al. (eds), Elsevier, Netherlands, pp.201-211.
247a. Callissano, P., Monaco, G., Castellani, L., Merchanti, D. and evi, A. (1978) Proc. Nat. Acad. Sci. U.S.A. 75, 2210.
248. Fiske, C.H. and Subba Row, Y. (1925) J. Biol. Chem.66, 375.
249. Jacobs, M. and Caplow, M. (1976) Biochem. Biophys. Res. Communs. 68, 127.